mercydiaz84
09.10.2019 •
Chemistry
Which substances have the same number of carbon atoms?

Solved
Show answers
More tips
- F Family and Home How to Raise a Genius? Discover the Secrets Here...
- P Philosophy How did the concept of module arise in computer science?...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- D Dating, Love, Relationships Why do we feel shame?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
Answers on questions: Chemistry
- M Mathematics Solve the equation for c M+3c=8...
- E English ..................................................................XD...
- E English Directions: Select one of the statements above that you feel you could write about. In the space below, write a paragraph that states your opinion (agree or disagree)...

Ответ:
Ответ:
The atoms whose ions have electronic configuration as are Iridium (Ir), Iron (Fe) and Rhenium (Re)
are Iridium (Ir), Iron (Fe) and Rhenium (Re)
Explanation:
An ion is formed when a neutral atom looses or gains electrons.
When an atom looses electrons, it results in the formation of positive ion known as cation.When an atom gains electrons, it results in the formation of negative ion known as anion.For the given elements:
Iridium: The electronic configuration of this element is![[Xe]6s^25d^7](/tpl/images/0476/0941/6f024.png) . The stable oxidation state of iridium is +4.
. The stable oxidation state of iridium is +4.
The electronic configuration of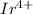 is
is ![[Xe]5d^5](/tpl/images/0476/0941/483d2.png)
Radon: The electronic configuration of this element is![[Xe]6s^25d^{10}6p^6](/tpl/images/0476/0941/63090.png) . This is a stable element.
. This is a stable element.
Iron: The electronic configuration of this element is![[Ar]4s^23d^6](/tpl/images/0476/0941/ed2f9.png) . The stable oxidation state of iron is +3.
. The stable oxidation state of iron is +3.
The electronic configuration of is
is ![[Ar]3d^5](/tpl/images/0476/0941/2060b.png)
Protactinium: The electronic configuration of this element is![[Rn]7s^26d^15f^2](/tpl/images/0476/0941/f2d20.png) . The stable oxidation state of protactinium is +3.
. The stable oxidation state of protactinium is +3.
The electronic configuration of is
is ![[Rn]7s^2](/tpl/images/0476/0941/e51f5.png)
Ytterium: The electronic configuration of this element is![[Kr]5s^24d^1](/tpl/images/0476/0941/e367b.png) . The stable oxidation state of ytterium is +3.
. The stable oxidation state of ytterium is +3.
The electronic configuration of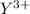 is
is ![[Ar]4s^23d^{10}4p^6](/tpl/images/0476/0941/5a2c9.png)
Rhenium: The electronic configuration of this element is![[Xe]6s^25d^5](/tpl/images/0476/0941/31aad.png) . The stable oxidation state of rhenium is +2.
. The stable oxidation state of rhenium is +2.
The electronic configuration of is
is ![[Xe]5d^5](/tpl/images/0476/0941/483d2.png)
Hence, the atoms whose ions have electronic configuration as are Iridium (Ir), Iron (Fe) and Rhenium (Re)
are Iridium (Ir), Iron (Fe) and Rhenium (Re)