QueenNerdy889
09.12.2020 •
Chemistry
(will mark brainliest) pls don’t scam - due today! these are my last points please answer! please answer the first and second one
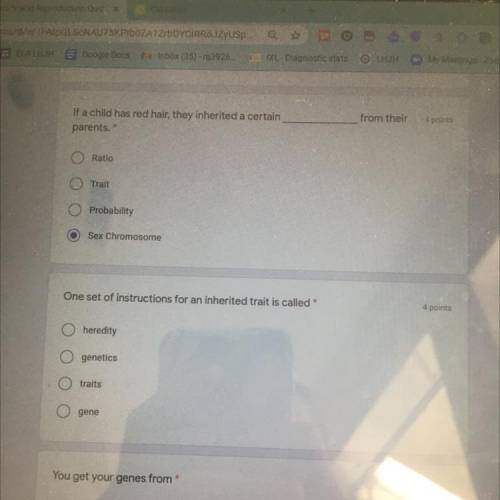
Solved
Show answers
More tips
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry Which of the following changes of state is exothermic evaporation? melting? or freezing?...
- C Chemistry Amaterial that is not easily scratched is _...
- C Chemistry What is true when a reaction has reached equilibrium? question 4 options: the reaction has stopped. the reaction is faster in the forward direction. the reaction is faster...
- C Chemistry Predict the product of Ag3N+Ba...
- C Chemistry Why does the OH molecule in alcohol have 1/2 positive and 1/2 negative charge?...
- C Chemistry Escriba la ecuación que le corresponde a la siguiente reacción y luego equilíbrarla por el método de tanteo: cobre + nitrato de plata, para dar nitrato de cobre (II) y plata...
- M Mathematics What is the range of the quadratic function?...
- H Health Suppression’s, or win/win or win/lose scenario, can create alignment of a systems member...
- M Mathematics The sum of 4 times a number and -3 added to 9 times the number...
- C Chemistry What volume of hydrogen will be predicted at stp by the reaction of 78.33g of aluminum with excess water...

Ответ:
18.02 grams
The average mass of one mole of H2O is 18.02 grams.
Explanation: we need to find the moles of water in 1 L of water or 1000mL of water. Taking the density of water to be 1g/mL, 1000mL of water= 1000g water.
Therefore, number of moles= given mass of water(1000)/molecular mass of water(18)
Moles =1000/18= 55.55
So total molecules in 55.55 moles= 55.55* 6.022*10^23 = 334.52*10^23 molecules
=3.34*10^25 molecules of water.
:)