brianabrady06
21.12.2019 •
Chemistry
Write a net ionic equation for the reaction that occurs when excess hydrochloric acid (aq) and potassium sulfite (aq) are combined. note: sulfites follow the same solubility trends as sulfates.
Solved
Show answers
More tips
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- H Health and Medicine At What Age Does a Person Stop Growing?...
- F Family and Home How to Choose a Name for Your Baby?...
- F Food and Cooking Discover the Health Benefits of Cedar Nuts...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- F Food and Cooking How to Make Sushi: A Step-by-Step Guide to Perfectly Rolled Delights...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- F Food and Cooking Аэрогриль: Все, что нужно знать для подготовки здоровой и вкусной пищи...
- F Food and Cooking The Health Benefits of Flaxseed oil...
Answers on questions: Chemistry
- H History as long as the reason of man continues fallible, and he is at liberty to exercise it, different opinions will be formed the diversity in the faculties of men, from which the...
- M Mathematics Can someone explain how to do this: If you put 10 in to the function f(x) = 2x, what would be the output? -- Im so lost-...
- H History This map shows that the silk road? 1- Crossed both african and asia 2- was located primarily in asia 3- followed a single route 4- started in khotan...

Ответ:
The net ionic equation for the given reaction is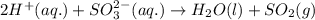
Explanation:
Net ionic equation of any reaction does not include any spectator ions.
Spectator ions are the ions which do not get involved in a chemical equation. It is also defined as the ions that are found on both the sides of the chemical reaction when it is present in ionic form.
The chemical equation for the reaction of hydrochloric acid and potassium sulfite is given as:
Ionic form of the above equation follows:
As, potassium and chloride ions are present on both the sides of the reaction, thus, it will not be present in the net ionic equation.
The net ionic equation for the above reaction follows:
Hence, the net ionic equation for the given reaction is written above.
Ответ: