Snowinvain
20.09.2019 •
Chemistry
Write the equilibrium constant expression for the following reaction in terms of concentrations of the components. (concentration equilibrium expressions take the general form: kc = [c]c / [a]a . [b]b. subscripts and superscripts that include letters must be enclosed in braces {}.) mgco3(s) equilibrium reaction arrow mgo(s) + co2(g)
Solved
Show answers
More tips
- C Computers and Internet Are there special gaming mice?...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- B Biology What is deficiency diseases...
- H History Free points free points for who ever finds it first Merry Christmas...
- E English Whats a comparison to the book and movie to the adventures of tom sawyer im doing an essay about it...
- M Mathematics In the diagram, N, O, and P are congruent. Find x....

Ответ:
The expression for equilibrium constant in terms of concentration is![K_c=[CO_2]](/tpl/images/0244/8923/0ff0c.png)
Explanation:
Equilibrium constant in terms of concentration is defined as the ratio of concentration of products to the concentration of reactants each raised to the power their stoichiometric coefficients. It is represented by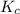
For a general chemical reaction:
The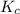 is written as:
is written as:
The concentration of pure solids and pure liquids are taken as 1.
For the given chemical reaction:
The expression for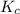 is:
is:
In the above expression, magnesium oxide and magnesium carbonate will not appear because they are present in solid state.
So, the expression for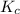 becomes:
becomes:
Hence, the equilibrium constant for the reaction is given above.
Ответ:
Carbon can react with oxygen to form carbon dioxide. Which of the following statements about this chemical change is true? ... Carbon and oxygen atoms are destroyed as new atoms are formed. Carbon and oxygen atoms have the same properties as molecules of carbon dioxide.