gonzaleze18
30.07.2020 •
Chemistry
Write the net ionic equation for any precipitation reaction that may be predicted when aqueous solutions of manganese(II) nitrate and sodium hydroxide are combined.
Solved
Show answers
More tips
- S Society and Politics What If There s War Tomorrow, What If We Go to War?...
- S Sport How to Do a Jumping Split...
- A Animals and plants What Do Terriers Eat?...
- F Food and Cooking Discover the Benefits and Properties of Dates...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- M Mathematics . The graph below shows the number of pies and the number of cakes that the students in the art club need to sell at the school bake sale in order to raise $150....
- H Health Which description best characterizes the jazz of the harlem renaissance?...
- M Mathematics Two models of the same compound are shown.In what way is Model A better than Model B?Model A shows the types of elements in the compound, but Model B does not.Model A shows the...
- M Mathematics H (2) = 5x + 2 Find x if h (x) = -13...
- H Health Your family recently adopted a dog from an animal shelter. initially, the dog seemed nervous and territorial, but after a few weeks, she became affectionate and calm. which of the...

Ответ:
Explanation:
Mn( NO₃ )₂ + 2Na OH = Mn( OH)₂ (s) ↓ + 2Na NO₃
Converting into ions
Mn⁺ + 2 NO₃⁻ + 2 Na⁺ + 2 OH⁻ = Mn( OH)₂ + 2 Na⁻ + 2 NO₃⁻
Cancelling out common terms
Mn⁺ + 2 OH⁻ = Mn( OH)₂
this is net ionic equation required.
Ответ:
Answer : The balanced chemical equation in a acidic solution is,
Explanation :
Redox reaction or Oxidation-reduction reaction : It is defined as the reaction in which the oxidation and reduction reaction takes place simultaneously.
Oxidation reaction : It is defined as the reaction in which a substance looses its electrons. In this, oxidation state of an element increases. Or we can say that in oxidation, the loss of electrons takes place.
Reduction reaction : It is defined as the reaction in which a substance gains electrons. In this, oxidation state of an element decreases. Or we can say that in reduction, the gain of electrons takes place.
Rules for the balanced chemical equation in acidic solution are :
First we have to write into the two half-reactions.
Now balance the main atoms in the reaction.
Now balance the hydrogen and oxygen atoms on both the sides of the reaction.
If the oxygen atoms are not balanced on both the sides then adding water molecules at that side where the less number of oxygen are present.
If the hydrogen atoms are not balanced on both the sides then adding hydrogen ion at that side where the less number of hydrogen are present.
at that side where the less number of hydrogen are present.
Now balance the charge.
The given chemical reaction is,
The oxidation-reduction half reaction will be :
Oxidation :
Reduction :
First balance the main element in the reaction.Oxidation :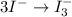
Reduction :
Now balance oxygen atom on both side.Oxidation :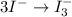
Reduction :
Now balance hydrogen atom on both side.Oxidation :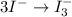
Reduction :
Now balance the charge.Oxidation :
Reduction :
The charges are balanced on both side of the reaction. Thus, added both equation, we get the balanced redox reaction.
The balanced chemical equation in acidic medium will be,