poniatowski23
06.07.2019 •
Chemistry
Xenon (xe) of mass 5.08 g reacts with fluorine to form 9.49 g of a xenon fluoride compound. what is the empirical formula of this compound?
Solved
Show answers
More tips
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
Answers on questions: Chemistry
- B Business Swift oil company is considering investing in a new oil well. it is expected that the oil well will increase annual revenues by $140,000 and will increase annual...
- L Law Which explains the connection between the law of demand and excess demand? the lo states that decreases in price leads to greater quantity demanded and limited supply,...
- H History How would you characterize the Eastern Front?...

Ответ:
mass of Xenon (Xe) = 5.08 g
mass of Fluorine (F) = 9.49 g - 5.08 g = 4.41 g
Determine the number of moles of each of the element in the compound.
moles of Xenon (Xe) = (5.08 g)(1 mol Xe / 131.29 g of Xe) = 0.0387 mols of Xe
moles of Fluorine (F) = (4.41 g)(1 mol F/ 19 g of F) = 0.232 mols of F
The empirical formula is therefore,
Xe(0.0387)F(0.232)
Dividing the numerical coefficient by the lesser number.
XeF₆
Ответ:
Approximately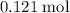 .
.
Explanation:
Balance the equationTo balance this equation, start by setting the coefficient of the most complex species to . For example,
. For example,  has more atoms in each of its formula unit than any other species in this reaction. Let the coefficient of
has more atoms in each of its formula unit than any other species in this reaction. Let the coefficient of
That formula unit of would include:
would include:
The other product, , contains neither
, contains neither  atoms nor
atoms nor  atoms. Therefore, the product side would include exactly
atoms. Therefore, the product side would include exactly
oneAtoms are conserved in a chemical reaction. The reactant side shall also include:
Among the reactants, is the only source of
is the only source of  atoms. Each formula unit of
atoms. Each formula unit of
Similarly, the coefficient of would be
would be  .
.
There would be exactly
 atom on the reactant side (from
atom on the reactant side (from  .) The products should also include
.) The products should also include
Hence the equation:
 .
.
Calculate n(Cu(NO₃)₂ (aq))The ratio between the coefficients of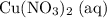 and
and 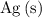 is:
is:
Rearrange this equation to obtain:
In other words,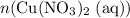 (the number of moles of
(the number of moles of 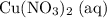 that is produced) can be found from
that is produced) can be found from  (the number of moles of
(the number of moles of 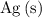 that is produced.)
that is produced.)
On the other hand,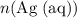 can be found from
can be found from 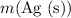 (the mass of
(the mass of 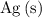 that is produced) using
that is produced) using  (the formula mass of
(the formula mass of  .)
.)
Look up relative atomic mass data of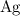 on a modern periodic table:
on a modern periodic table:
Therefore, the relative atomic mass of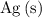 would be:
would be:
Calculate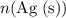 :
:
Calculate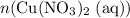 :
: