mothertrucker2828
18.03.2020 •
Chemistry
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca in the rock. You dissolve the limestone in hydrochloric acid, which gives a solution of calcium chloride. Then you precipitate the calcium ion in solution by adding sodium oxalate, Na2C2O4. The precipitate is calcium oxalate, CaC2O4. You find that a sample of limestone weighing 128.3 mg gives 140.2 mg of CaC2O4. What is the mass percentage of calcium in the limestone
Solved
Show answers
More tips
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry tissues maples and white pines are two different tree species that often grow side by side in the aderondack mountains which statement concerning these trees is correct?...
- C Chemistry Please help it is very needed...
- C Chemistry HELLLP PLEASEE hElPpPp ILL MaRk You AS Brainlister...
- C Chemistry When the nuclide polonium-218 undergoes alpha decay: a. The name of the product nuclide is . b. The symbol for the product nuclide is . Write a balanced nuclear equation...
- C Chemistry 1. Two hypothetical elements in the periodic table have different characteristics as shown in the following table. ELEMENT, ELECTRONEGATIVITY, TYPE OF ION, ELECTRICAL...
- M Mathematics Arectangle has an area of 7 7/8 square inches and a length of 3 1/2 inches. the rectangle s width is inches. write your answer as a mixed number in lowest terms....
- M Mathematics What are the mean and standard deviation of the sampling distribution of x bar in random samples of n=100...
- E English What are the three pieces of evidence simon schema gives to support his central claim in the article the shakespearean shakedown ? in grade 8 module 2 a midsummer night...
- E English Using sources imagine that you re at your family dinner table, with all of your family members around you. everyone is casually eating and talking amongst themselves...
- M Mathematics Y=-4(x+3)-2 as a slope intercept function...

Ответ:
34.15% is the mass percentage of calcium in the limestone.
Explanation:
Mass of precipitate that is calcium oxalate = 140.2 mg = 0.1402 g
1 mg = 0.001 g
Moles of calcium oxalate =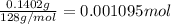
1 mole of calcium oxalate have 1 mole of calcium atom.
Then 0.001095 moles of calcium oxalate will have 0.001095 moles of calcium atom.
Mass of 0.001095 moles of calcium :
0.001095 mol × 40 g/mol = 0.04381 g
Mass of sample of limestone = 128.3 mg = 0.1283 g
Percentage of calcium in limestone:
34.15% is the mass percentage of calcium in the limestone.
Ответ:
This is because you know that these are the same objects, since it’s asking to find the scale factor. You are able to correlate AB to EF, and that shows a scale UP of 1.5, from 2 to 3. With this scale factor of 1.5, we know that EH will be 1.5 times bigger than AD. So 1.5 x 5 is equal to 7.5.