genyjoannerubiera
31.07.2019 •
Chemistry
You need to produce a buffer solution that has a ph of 5.31. you already have a solution that contains 10. mmol (millimoles) of acetic acid. how many millimoles of acetate (the conjugate base of acetic acid) will you need to add to this solution? the pka of acetic acid is 4.74.
Solved
Show answers
More tips
- H Health and Medicine Why do our Joints Crack?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry 2) Explain how two students could perform chromatography on the same mixture, the colored samples all move different distances, yet they can correctly identify the component...
- C Chemistry The concentration of alcohol in adult beverages is typically expressed as proof. Proof is simply double the % by volume (% v/v). Assuming that vodka is a solution of...
- C Chemistry TVINN Assertion: Naphthalene,camphor, iodine, ammoniumchloride are some commonexamples of the substanceswhich undergo sublimation.Reason: All solids are firstconverted...
- C Chemistry Jelaskan kelebihan reka bentuk akuaponik...
- C Chemistry Frinken high and bored join no rude stuff hot boys welcome idrc for girls but your welcome too I ll show what i look like I m a girl and 18 https://pad let.com/alexis534/4f5e9tkbtpurlwht...
- C Chemistry Opinion on the usage of identification of amine in daily life (medical, agriculture,etc)...
- C Chemistry In chemistry class, a student conducted an investigation on the factors that affect the solubility of copper (II) sulfate (CUSO) in water. The data were recorded in the...
- C Chemistry What two things combine with oxygen to form ground-level ozone? A) NOx and ODS B) CFCs and halons C) NOx and VOC D) HFCs and HCFCs...
- C Chemistry HELP ME PLEASE Which is true about the relationship between the grain size of the sediment and the permeability of the sediment? A. As the grain size of the sediment...
- C Chemistry 1) Calculate the temperature change for mercury if 160 g of the metal absorbs 1500 J of heat energy. Mercury s specific heat is 0.14 J/g°C. 2) How much heat is given...

Ответ:
37 mmol of acetate need to add to this solution.
Explanation:
Acetic acid is an weak acid. According to Henderson-Hasselbalch equation for a buffer consist of weak acid (acetic acid) and its conjugate base (acetate)-
Here pH is 5.31,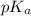 (acetic acid) is 4.74 and number of mmol of acetic acid is 10 mmol.
(acetic acid) is 4.74 and number of mmol of acetic acid is 10 mmol.
Plug in all the values in the above equation:
or, mmol of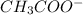 = 37
= 37
So 37 mmol of acetate need to add to this solution.
Ответ:
25 mL
Explanation: