You wish to add 5 mg/l naocl as cl2 to a solution in a disinfection test, and you have a stock solution (household bleach) that contains 5% naocl by weight. assuming that the density of the stock solution is 1.0 g/ml, how many milliliters of bleach should you add to each liter of test solution?
Solved
Show answers
More tips
- O Other What is the oldest joke ever told?...
- F Food and Cooking How to Make Lazy Cabbage Rolls? Simple Steps to a Delicious Dish...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
Answers on questions: Chemistry
- C Chemistry What would be the mass, in grams, of 1.806 x 1024 molecules of carbon monoxide? YOU MUST SHOW ALL OF THE STEPS AND YOU MUST DO THE PROBLEM JUST AS ILLUSTRATED. INCLUDE UNITS!...
- C Chemistry A 1 L sample from Tempe Town Lake has a pH of 3.57. Exhaust from cars on nearby highways often mixes with rain to form HNO3. Use this information to answer the following questions....
- C Chemistry What number represents a mole? 5 1 4 14 8 6 19...
- C Chemistry The electronic configuration of an element is 1s2 2s2 2p6 3s2 3p1 . An atom of this element form an ion that will have charge (A) +1 (B) +2 (C) +3 (D) -1...
- C Chemistry If two atoms of the same element bond together to form a diatomic molecule the bond between them would be considered a: a.polar covalent bond b.ionic bond c.nonpolar covalent bond...
- C Chemistry Draw the Lewis structure for the SeH2 molecule....
- C Chemistry How do we know the specific details of Lewis and Clark s expedition out West?...
- C Chemistry The active component in one type of calcium dietary supplement is calcium carbonate. A 1.2450 g tablet of the supplement is added to 65.00 mL of 0.4984 M HCl(aq) and allowed to...
- C Chemistry You are asked to test the biochemical oxygen demand of a stream in your town. After observing the BOD over a week, you calculate that the water has a BOD of 150 mg of oxygen, which...
- C Chemistry A 1.10 g sample contains only glucose and sucrose. When the sample is dissolved in water to a total solution volume of 25.0L, the osmotic pressure of the solution is 3.78 atm at...

Ответ:
-
0.1 ml of bleach should be added to each liter of test solution.
Explanation:-
Let the volume of bleach to be added is B ml.
Density of stock solution = 1.0 g/ml
Mass of stock solution = Volume of stock x density of stock
= B ml x 1.0 g/ml
= B g
Amount of NaOCl in this stock solution = 5% of B g
= x B g
x B g
= 0.05 B g
Now each test solution must be added 5 mg/l NaOCl.
Thus each liter of test solution must have 5 mg.
Thus 0.05 B g = 5 mg
= 0.005 g
B =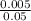
= 0.1
Thus 0.1 ml of bleach should be added to each liter of test solution.
Ответ:
:
Answer is 28.
all you have to do is draw a model of 4 atoms and add each molecule to find the answer of 28!