Which of the following sentences is a declarative sentence?
a. in the story, sylvia climbed a tall pine tree.
b. was climbing it a very difficult task?
c. what sort of tree did she climb?
d. to find out, just read the story.
Solved
Show answers
More tips
- W Work and Career Мерчендайзинг – все, что нужно знать...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
Answers on questions: English
- H History Where did most enslaved people work in Louisiana during the antebellum era? textile mills family farms city households large plantations...
- M Mathematics Find the area of the circle shown below if the radius is 13 inches. Use 3.14 for π. Use the formula π x R^2 A) None of these answers B)2,122.64 inches C)530.66 inches D)20.41...
- H History What are the events that divided the nation and led to civil war...
- E English Read the following paragraph and select the correct answer. a prison taint was on everything there. the imprisoned air, the imprisoned light, the imprisoned damps, the imprisoned...

Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of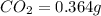
Mass of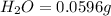
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,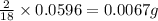 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =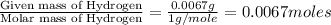 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =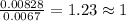
For Hydrogen =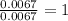
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.