I can't-
This person-

Solved
Show answers
More tips
- F Food and Cooking Kick Your Taste Buds Into Gear with This Authentic and Delicious Recipe for Solyanka...
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
Answers on questions: History
- B Biology Which of the following is the best example of an open system? a) an insulated gas cylinder b) a car engine c) our universe d) a greenhouse...
- H History How did these weapons transform warfare? what resulted?...
- M Mathematics Bradly completed 0.8 of his homework and Max completed 8% of his homework. Who completed more of their homework? How do you know? Show all work and explain your answer. ...
- M Mathematics Find the area of the polygon....
- E English How has lord of the flies influenced popular culture?...

Ответ:
The unit cell edge length for the alloy is 0.405 nm
Explanation:
Given;
concentration of Ag,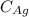 = 78 wt%
= 78 wt%
concentration of Pd, = 22 wt%
= 22 wt%
density of Ag = 10.49 g/cm³
density of Pd = 12.02 g/cm³
atomic weight of Ag,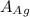 = 107.87 g/mol
= 107.87 g/mol
atomic weight of iron,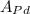 = 106.4 g/mol
= 106.4 g/mol
Step 1: determine the average density of the alloy
Step 2: determine the average atomic weight of the alloy
Step 3: determine unit cell volume
for a FCC crystal structure, there are 4 atoms per unit cell; n = 4
Step 4: determine the unit cell edge length
Vc = a³
Therefore, the unit cell edge length for the alloy is 0.405 nm