juanchylj
10.02.2020 •
Mathematics
A first order decomposition reaction has a half-life of 64yr. What percentage of the original amount is left after 135years. (give answer to 1 decimal place)?
Solved
Show answers
More tips
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...

Ответ:
23.2%
Step-by-step explanation:
First we need to find how many half lives 135 years is:
135 ÷ 64 = 2.109375
Now we divide 100% by 2 to the power of the number of half lives. This is how you calculate the percentage of the original amount left. If you have the original mass, you can substitute that in instead of 100%:
100 ÷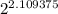 = 23.1747390635
= 23.1747390635
To 1 decimal place it is 23.2%
Ответ:
345
Step-by-step explanation: