21hendlill
27.02.2021 •
Mathematics
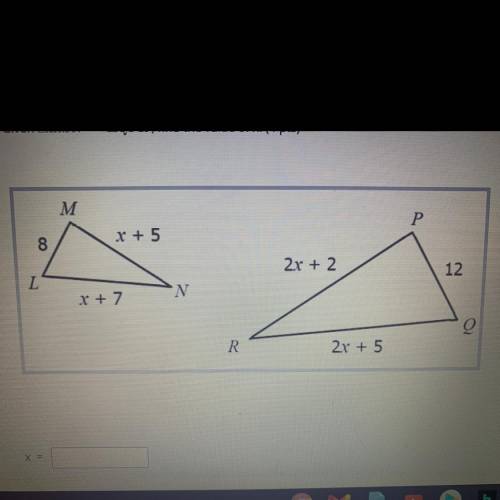
Given LMN ~ QPR, find the value of x
Solved
Show answers
More tips
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to Braid Hair with a Plaiting Machine: Tips and Recommendations...
- C Computers and Internet Where did torrents.ru move to?...
- H Health and Medicine How to Get Pregnant Faster?...
- A Animals and plants Money Tree Care Secrets: How to Keep Your Plant Thriving...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- H Health and Medicine What was the Invention of Viagra?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
Answers on questions: Mathematics
- M Mathematics I need help again please...
- M Mathematics Part 2. The transformation shown is a reflectiontrue or false...
- M Mathematics Use the distributive property to write an expression that is equivalent to 27ײ - 42x + 12...
- M Mathematics Triangle ABC is dilated using a scale factor of 1/3 to form a triangle A B C . The new coordinates are A (1,-1),B (4,-1) and C (1,-5). What are the ORIGINAL coordinates...
- M Mathematics What is 1 whole and 1/4 + 3/4...
- M Mathematics Please Help me I need to finish this worksheet Given: AB ∥ CD AC ∥ BDProve: 1 ≅ 4...
- M Mathematics Order these numbers from least to greatest. 7.008, 7.3, 7.3071, 7.08...
- M Mathematics I need help thank you...
- M Mathematics If 32 horses consume 112 kg of grass in a certain period, how much grass will be consumed by 11 horses during the same period?...
- M Mathematics What is the solution to -1.6 b - (-4.4) A:2.8 bB: -6.0 bC: 6.0 bD. 2.8 b...

Ответ:
The answer is x=11
Step-by-step explanation:
Ответ:
6.64*10^(-9) moles of oxygen.
Step-by-step explanation:
If we have N molecules, the number of moles is given by:
n = N/Nₐ
where Nₐ is the Avogadro's number:
Nₐ = 6.02214076*10^23
In 2.000*10^15 molecules of sulfur dioxide, we have:
n = (2.000*10^15)/(6.02214076*10^23) = 3.32*10^(-9) moles of sulfur dioxide.
Now, sulfur dioxide has two oxygens and a single sulfur.
Then a mole of sulfur dioxide has two moles of oxygen and a single mol of sulfur.
Then in 3.32*10^(-9) moles of sulfur dioxide, we have twice that amount moles of oxygen, or:
2*3.32*10^(-9) = 6.64*10^(-9) moles of oxygen.