alleyeverett031
26.04.2021 •
Mathematics
Hexagon A’B’C’D’E’F’ is the image of the hexagon ABCDEF
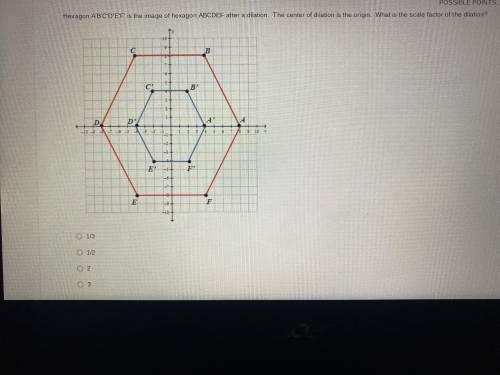
Solved
Show answers
More tips
- C Computers and Internet Protect Your Computer: How to Set a Password on Your Device...
- W Work and Career How to behave at an interview? Tips from an HR specialist...
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Mathematics
- M Mathematics If the dot product of two non zero vectors, v1 and v2 is zero, what does this tell us?...
- M Mathematics Please answer it’s due asap!...
- M Mathematics Jack had 3bags of golf balls with b balls in each bag; then his friend gave him 6 more golf balls. How many golf balls does Jack have now?...
- C Computers and Technology A(n) is a common output device for hard copy. a. laser printer b. plasma display c. compact disc d. optical character reader...
- M Mathematics List all the factors of the numbers: a. 40 and b. 3 Answer fast...
- M Mathematics Solve the equation (If possible please show work)...
- H History What was the strictest condition Johnson required of Southern states?...

Ответ:
Option (c) is the correct answer.
Explanation:
Bronsted-Lowry stated that acids are the species which donate a proton whereas bases are the species which accept a proton.
Also, a strong acid results in the formation of a weak conjugate base and a weak acid results in the formation of a strong conjugate base.
Similarly a strong base results in the formation of a weak conjugate acid and a weak base results in the formation of a strong conjugate base.
The reaction for hydrofluoric acid and acetic acid is as follows.
Thus, hydrofluoric acid being a strong acid gives a weak conjugate base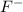 whereas acetic acid being a weak acid gives strong conjugate base
whereas acetic acid being a weak acid gives strong conjugate base 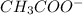 .
.
Hence, the conjugate base of hydrofluoric acid is weaker than that of acetic acid.