mshurney
12.05.2021 •
Mathematics
Please I have 15 min

Solved
Show answers
More tips
- P Philosophy What is Something for you?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Mathematics
- M Mathematics Can anyone find the pdf for glencoe pre-algebra chapter 11 resource masters, ....
- M Mathematics Amanda has 15 coins in her pocket. of these, 8 are quarter. what is ratio of quarters to coins in amanda s pocket? i...
- M Mathematics X2 + 12 = 48 a. 36, -36 b. 6,-6 c. 18,-18...
- M Mathematics Caillans pocket money increased from $15 per week to $20 per week. how much has caillans pocket money increased? what percentage increases in this?...
- M Mathematics If it takes six men six days to dig six holes, how long will it take one man to dig half a hole?...
- M Mathematics Larry paid 11.20 for four gallons of gas how much was each gollon of gas...
- M Mathematics What property allows us to determine the congruency of the two triangles in the diagram? a. sss b. asa c. sas d. hl...
- M Mathematics There are 207 students in 9 classes at this rate how many are in 6 classes...
- M Mathematics write the following in function notation. y = 3x - 10...
- M Mathematics What is the simplified fraction for 1 1/2 times 5 5/6?...

Ответ:
False
Step-by-step explanation:
Ответ:
False
Step-by-step explanation:
Ответ:
The sample is 13414 years old.
Step-by-step explanation:
Expression for rate law for first order kinetics is given by:
where,
k = rate constant
t = age of sample
a = let initial amount of the reactant= 100
a - x = amount left after decay process=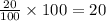
a) for completion of half life:
Half life is the amount of time taken by a radioactive material to decay to half of its original value.
b) for completion of 20% of reaction
The sample is 13414 years old.