h2o12
01.02.2022 •
Mathematics
Please I need this answer fast
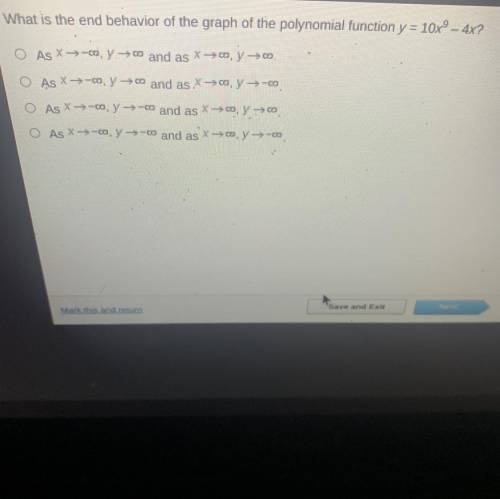
Solved
Show answers
More tips
- G Goods and services How to Choose a Video Camera: Tips from Professionals...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Mathematics
- M Mathematics If 75% of 960 = 30a, then a =...
- M Mathematics Mario and Luigi together have a total of 84 coins. Luigi has 10 more coins than Mario. How many coins does Luigi have?...
- M Mathematics HELP ASAP WHAT ONE IS IT...
- M Mathematics Which point is a solution to the system x2 + y2 ≤ 25 and x y2 – 2? (–5, 0) (–2, 0) (5, 0) (2, 2)...
- M Mathematics NEEED HELP ASAP! c2 =?...
- M Mathematics Find 0.35 percent of 80...
- M Mathematics What are the x- and y-intercepts for the graph of 3x -2y =6...
- M Mathematics PLZ, help me with this question....
- M Mathematics I need help on this problem...
- M Mathematics Are 2 + 5x and 5x + 2 equivalent?...

Ответ:
c x approaches -infinity and y approaches -infinity and vice versa
Step-by-step explanation:
graphed on calculator
Ответ:
The chemical reaction produces a new substance with new and different physical and chemical properties. Matter is never destroyed or created in chemical reactions. The particles of one substance are rearranged to form a new substance. The same number of particles that exist before the reaction exist after the reaction.
hope this helps
Step-by-step explanation: