jazzy9927
26.11.2019 •
Mathematics
The radius of the aluminum atom is 143pm. the radius of the aluminum atom is 54pm. by what percentage did the radius change as the ion formed?

Solved
Show answers
More tips
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- P Philosophy How to Develop Extrasensory Abilities?...
Answers on questions: Mathematics
- M Mathematics Helppppppppppppp helppppppp...
- M Mathematics Ineed to solve the inequality but the equal sign is confusing me, what do i do?...
- M Mathematics Ayudenmeee. Por favor...
- M Mathematics Whats the sum of 7/2/5 yards and 3/5 yards...
- C Chemistry What atom has the larger atomic radius ca or ni ?...

Ответ:
There was 62.23% change in radius as the ion formed.
Step-by-step explanation:
Given
Radius of Aluminium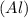 atom = 143 pm
atom = 143 pm
Radius of Aluminium atom = 54 pm
atom = 54 pm
Change is the radius = Radius of Aluminium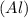 atom - Radius of Aluminium
atom - Radius of Aluminium  atom = 143 -54 = 89
atom = 143 -54 = 89
Now to find % Change is the radius we will divide Change is the radius by Radius of Aluminium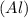 atom and multiply by 100 we get
atom and multiply by 100 we get
% Change is the radius =
Hence there was 62.23% change in radius as the ion formed.
Ответ:
its 90% if you work it out
Step-by-step explanation:
I hope this is the answer