snurpeisov
28.06.2020 •
Physics
A gas fills the right portion of a horizontal cylinder whose radius is 5.33 cm. The initial pressure of the gas is 2.29 x 105 Pa. A frictionless movable piston separates the gas from the left portion of the cylinder that is evacuated and contains an ideal spring, as the drawing shows. The piston is initially held in place by a pin. The spring is initially unstrained, and the length of the gas-filled portion is 18.9 cm. When the pin is removed and the gas is allowed to expand, the length of the gas-filled chamber doubles. The initial and final temperatures are equal. Determine the spring constant of the spring.
Solved
Show answers
More tips
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- F Food and Cooking How to Make Thin Pancakes: Recipe and Tips...
- S Style and Beauty Is Hot Scissor Haircutting Beneficial or Dangerous?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking Is Bacon Good for You?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
Answers on questions: Physics
- M Mathematics A closed box has a square base with side length l feet and height h feet. Given that the volume of the box is 29 cubic feet, express the surface area of the box in terms...
- C Chemistry Worldwide, the three main tidal patterns that exist are diurnal tides, tides and mixed tides...
- E English Read the following scenario. Shonda is a high school junior who works weekends delivering laundry at a nursing home. She hopes that before she graduates from high school...

Ответ:
k = 5406.88 N/m
Explanation:
Since the initial and final temperatures are constant while the gas expands, the expansion of the gas obeys Boyle's law
Initial gas pressure,
Let the initial gas volume = V
= V
Volume = Length * Area
Since the cross sectional area does not change and the length of the gas-filled chamber is said to double, then the volume also doubles.
The final gas pressure,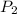 will be gotten from the Boyle's law equation
will be gotten from the Boyle's law equation
The Cross Sectional Area,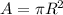
R = 5.33 cm = 0.0533 m
The force exerted on the piston, F = P₂ A
F = 1.145 * 10⁵ * 0.0089
F = 1021.9 N
To get the spring constant, use Hooke's law
F = k Δx
Where Δx = l₂ - l₁
Δx = 2l - l = l ( since the length of the chamber doubles on expansion)
Δx = 18.9 cm = 0.189 m
F = k Δx
1021.9 = k * 0.189
k = 1021.9/0.189
k = 5406.88 N/m
Ответ: