lanakay2006
01.03.2020 •
Physics
A. How many calories are needed to raise the temperature of 1 gram of water by 1 °C?
B. How many calories are needed to melt 1 gram of ice at 0 °C?
C. How many calories are needed to vaporize 1 gram of boiling water at 100 °C?
Solved
Show answers
More tips
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Physics
- S Spanish Listen to the audio and then answer the following question. Feel free to listen to the audio as many times as necessary before answering the question.What does the doctor say...
- M Mathematics Drag the answer into the box to match each relation 0 4 4+ 3+ 2+ 1+ 3 2 1 1 -27 Domain: {-3, -1, 0,3} Domain {-3, -2.0, 2) Domain (-2,0,2,0) Range: {-1, 0, 1, 2, 3) Range: {0,...
- P Physics - As you ride in a car, both you and the car are moving at a constant speed. If the car brakes suddenly, you feel your body pulled forward. Why? A. Your body is in motion, and...

Ответ:
A) 1 cal
B) 80 cal
C) 540 cal
Explanation:
A)
The amount of heat energy needed to raise the temperature of a certain mass of a substance is given by
where
m is the mass of the substance
C is the specific heat capacity
In this problem:
m = 1 g is the mass of water
So, the heat needed is
B)
For a solid substance at its melting point, the amount of heat needed to melt completely the substance is given by
where
m is the mass of the substance
In this problem:
- The ice is already at melting point, 0 °C
- Mass of the ice: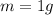
- Specific latent heat of fusion of ice: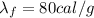
So, the heat needed is
C)
For a liquid substance at its boiling point, the amount of heat needed to boil completely the substance is given by
where
m is the mass of the substance
In this problem:
- The water is already at boiling point, 100 °C
- Mass of the water: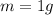
- Specific latent heat of vaporization of water:
So, the heat needed is
Ответ:
EXPLANATION:
E= Force x displacement
69=F(42)
69/42 = F
F=1.64 N
Answer : 1.6N