mzyjohnson47
18.08.2020 •
Physics
An ideal gas, initially at a pressure of 11.2 atm and a temperature of 299 K, is allowed to expand adiabatically until its volume doubles.
Required:
What is the gas’s final pressure, in atmospheres, if the gas is diatomic?
Solved
Show answers
More tips
- W Work and Career Secrets of Punctuality: How to Learn to Never Be Late?...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Physics
- H History How did statehood affect the political development of louisiana? a. it successfully blended louisiana’s traditional system of royal rule and american democracy. b. it...
- H Health Toby wants to gain experience in child development. toby chooses to enter a month-long certificate program after high school and begins working as a teacher s aide in...
- G Geography Mechanical weathering may occur by means of A. Plant roots. B. temperature changes. C. Frosty action. D. All of the above . E. None of the above....
- M Mathematics Find the slope of the line that passed through (5,8) and (1,13)...

Ответ:
The pressure is
Explanation:
From the question we are told that
The initial pressure is
The temperature is
Let the first volume be Then the final volume will be
Then the final volume will be 
Generally for a diatomic gas
Here r is the radius of the molecules which is mathematically represented as
Where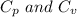 are the molar specific heat of a gas at constant pressure and the molar specific heat of a gas at constant volume with values
are the molar specific heat of a gas at constant pressure and the molar specific heat of a gas at constant volume with values
=>
=>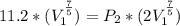
=>![P_2 = [\frac{1}{2} ]^{\frac{7}{5} } * 11.2](/tpl/images/0723/9998/ba19a.png)
=>
Ответ: