yourmumsanoodle
26.09.2019 •
Physics
Carbon dioxide (co2) gas within a piston–cylinder assembly undergoes a process from a state where p1 = 5 lbf/in.2 , v1 = 2.5 ft3 to a state where p2 = 20 lbf/in.2 , v2 = 0.5 ft3 . the relationship between pressure and volume during the process is given by p = 23.75 − 7.5v, where v is in ft3 and p is in lbf/in.2 . determine the work for the process, in btu.
Solved
Show answers
More tips
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- L Leisure and Entertainment How to Learn to Draw Graffiti: Tips for Beginners...
- G Goods and services How to sew a ribbon: Tips for beginners...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...
- H Health and Medicine Novomin: What is it and how to use it?...
- A Art and Culture Attention, the Final Episode of Margo is Almost Here!...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- S Style and Beauty How to Break in New Shoes: 7 Simple Methods...
Answers on questions: Physics
- E English In the text, the author discuss how students can make their time in college matter. How would you go about taking control of your education in college? Consider things...
- H Health Rajakr2103 be aware of him yaall child preadtor on the loose do not join any zoom callsis taking these down one by one sum one reports them STAY SAFE WOMAN...
- B Biology Which type of dna technology is used to caused bacteria to produce human insulin? a. recombinant dna b. pcr c. nuclear transfer d. gel electrophoresis...
- M Mathematics Calcula la energía de un ave de 2 kilogramos de masa que vuela a 150 m de altura con una velocidad de 40 m sobre segundo ...

Ответ:
work 24.31 ft³/in²
Explanation:
The work in thermodynamic processes is given by a relationship between pressure changes and volume differences as follows:
W = -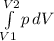
In this exercise they give the pressure ratio P = 23.75 -7.5 V, so we can evaluate the integral between points two V1 = 2.5 ft3 and V2 = 0.5 ft3
W = - [23.75 V -7.5 V² / 2]
W = - [23.75 (0.5 -2.5) - 7.5 (0.5² - 2.5²) / 2]
W = - [-47.5 + 23.19] BTU
W = 24.31 ft3 / in2 = 24.31 BTU
Note that the work is positive since work is done on the gas to compress it.
Ответ:
Sodium: C) Malleable, soft, and shiny
Silicon: B) Has properties of both metals and nonmetals
Bromine: D) Highly reactive gas
Argon: A) Nonreactive gas
Hope this helps :)