zombiechick180
01.07.2020 •
Physics
Four cubes of the same volume are made of different materials: lead (density 11,300 kg/m3), aluminum (density 2700 kg/m3), wood (density 800 kg/m3), and Styrofoam (density 50 kg/m3). You place the cubes in a large container filled with water.Rank the buoyant forces that the water exerts on the cubes from largest to smallest.Rank from largest to smallest. To rank items as equivalent, overlap them.
Solved
Show answers
More tips
Answers on questions: Physics
- C Computers and Technology (07 MC)Mary was chosen for the starring role in the school play. Her parents do not want her to participate because they worry her grades will suffer. What should Mary do?...
- S Social Studies If your single comment on this question something cool bout utah Utah is the second-driest state in the United States after Nevada. On average, Utah has about 300 sunny...
- M Mathematics Over the summer, Emily works a total of 15 hours a week. Each shift, she leaves with 30 dollars. But this week emily worked a total of 19 hours, how much money did she leave...
- M Mathematics Can you find the volume of a cone if you don’t know the height or radius?...
- M Mathematics What is the y-intercept of the graph of the line given by the equation 3y + 4x = 15? 05 04 03 0-4...

Ответ:
Lead > Aluminium > Wood > Styrofoam
Explanation:
Buoyant force is described by the Archimedes's Principle, which states that buoyant force is equal to the weight of the fluid displaced by the submerged object. By Newton's Laws, the buoyant force is represented by the following equation of equilibrium:
Where:
Let suppose that volume of the cube is known. Given that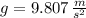 , the buoyant force is computed for each material:
, the buoyant force is computed for each material:
Lead ( )
)
Aluminium ( )
)
Wood (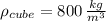 )
)
Styrofoam (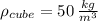 )
)
Therefore, the buoyant forces that the water exerts on the cubes from largest to smallest corresponds to: Lead > Aluminium > Wood > Styrofoam.
Ответ:
Graphite is great lubricant because each carbon atom forms weak covalent bonds with three other carbon atoms.
Diamond and graphite both are entirely made up of carbon but both the elements are completely different because of the bonding of carbon atoms. In graphite the carbon atoms form weak covalent with the other three carbon atom due to which there is less or no force between the atoms, and makes the graphite very soft and slippery, and graphite act as lubricant.