camballard3848
16.07.2019 •
Physics
Gas pressure within a fixed container will increase when 1) gas is cooled 2) gas is heated 4) gas volume changes 5) mass of a gas is increased
Solved
Show answers
More tips
- F Food and Cooking How to Determine Healthy, Nutritious Food for Yourself?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Physics
- P Physics Metal fins cool a motorcycle by heat to the outside...
- P Physics Which factors improve soil fertility? select the three correct answers. A:nitrogen B:horizons C:manure D:humus E:bedrock...
- P Physics The area around a magnet containing all of magnetic Lines of force is called...
- P Physics You are on a 5th story balcony, 12.5m above the sidewalk. Your friend is walking at 2.50 m/s north along the sidewalk. Where should your friend be in relation to the impact...
- P Physics Which of the following properly describes motion by comparing it to a reference point? A.the hands of a clock compared to the numbers on the clock. B.The distance of a race...
- H History Which cause of ww1 had the most impact starting the war?...
- M Mathematics Is this answer correct? show the steps...
- S Social Studies Why are there fewer conflicts between the executive branch and the legislative beach in a parliament...
- E English Dramatic irony is a literary device where the audience understands something that characters do not. which statement best describes an effect of dramatic irony? . a. it reveals...
- B Biology 2. the global winds in the first map generally travel in either a clockwise or counterclockwise direction. look at the global winds and compare the general direction of flow...

Ответ:
2) gas is heated
Explanation:
In this problem, we have a gas in a fixed container (so, the volume of the gas does not change). Let's see what happens in the different situations listed in the question.
1) Gas is cooled. Since the volume if fixed, the work done by the gas is zero: This means that the change in internal energy of the gas is equal to the heat exchanged:
This means that the change in internal energy of the gas is equal to the heat exchanged: 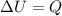 . However, since the gas is cooled, this means that Q is negative (heat is removed from the gas), so
. However, since the gas is cooled, this means that Q is negative (heat is removed from the gas), so 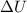 is negative, so the temperature of the gas, T, is decreased. Now if we look at the ideal gas equation
is negative, so the temperature of the gas, T, is decreased. Now if we look at the ideal gas equation
We see that since V, n and R remain constant, a decrease in T corresponds to a decrease in p: so, the pressure of the gas decreases.
2) Gas is heated. If we apply exactly the same reasoning of point 1), we see that when the gas is heated, its temperature increases: therefore, according to the ideal gas equation, its pressure must increase as well.
3) Gas volume change. By looking again at the ideal gas equation:
We see that in this case we don't have enough information to determine how does the gas pressure change. In fact, we don't know whether the volume V increases or decreases, so we cannot determine how p changes.
5) mass of a gas is increased. An increase in the mass of the gas corresponds to an increase in the number of moles, n. However, in the ideal gas equation, we don't know if the temperature of the gas, T, remains the same or not: therefore, we don't have enough information to tell how does p change.
Ответ:
Why do deep sea divers or high altitude fliers wear special suits? A: Humans are used to only a specific range of pressures. … Deep sea divers will have an atmospheric diving suit which block the heavy pressure of the water around them, and keep a decent pressure inside.
Explanation: