queenkimm26
22.01.2021 •
Physics
In a heat engine, if 500 J of heat enters the system, and the piston does 300 J of work.
What is the final internal (thermal) energy of the system if the initial energy is 1,500 J?
200 J
800 J
1,300 J
1,700 J
Solved
Show answers
More tips
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
Answers on questions: Physics
- H History An electrician needs 3 rolls of electrical wire to wire each room in a house. How many rooms can he wire with 1/2 of a roll of wire? Simplify your answer and write it as...
- E English Read the passage from “the lottery, part 1” that describes the moment mr. summers arrives. when he arrived in the square, carrying the black wooden box, there was a murmur...
- H History Look at the map. which ecosystems show where early civilizations developed because the area allowed people to grow crops even during times with little rainfall? select...
- E English Which sentence correctly uses an appositive phrase A.My Father waved as he dropped us off at school B. My father, a huge baseball fan, taught me how to throw when I was...

Ответ:
The final answer is 1,700 J
Explanation: According to first law of thermodynamics:
=Final energy-initial energy=Change in internal energy
q = heat absorbed or released
w = work done by or on the system
w = work done by the system= {Work done by the system is negative as the final volume is greater than initial volume}
q = +500J {Heat absorbed by the system is positive}
w = work done by the system = -300J
U2- 1500J= + 500J --300J
U2= 1700J
Ответ:
1)H=714 Watts of energy is given out.
2)614.34 KCal energy is burnt in an hour in the room
3)In freezing conditions, 1337.09 KCal is spent in an hour.
Explanation:
The formula for thermal conductivity states that the rate of heat loss H is as follows:
H= where,
where,
K=Coefficient of thermal conductivity
A=Area of Cross Section
H=Rate of heat loss
x=Thickness of the material
For first case, =17°C or 17°F
=17°C or 17°F
K for fat= 0.21
A=2
x=0.01 m
H=
H=714 Watts of energy is given out.
2) ×
×
Energy= 714 × 60 × 60=2570400 J
2570400 J=614.3403442 KCal
Therefore, 614.3403442 KCal energy is burnt in an hour.
3) Only the temperature difference becomes 37°C from 17°C , rest everything remains the same, therefore the energy will also vary due to the temperature factor and more energy will be spent as in freezing climate °C
°C
E=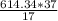 =1337.09 KCal
=1337.09 KCal
In freezing conditions, 1337.09 KCal is spent in an hour.