liloleliahx2
10.12.2019 •
Physics
The half-life for the α decay of uranium is 4.47 × 109 yr. determine the age (in years) of a rock specimen that contains 55.5% of its original number of atoms.
Solved
Show answers
More tips
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Physics
- E English Question 4(Multiple Choice Worth 4 points) (MC) You see an ad in the movie theatre for a new computer game. There is an American eagle proudly flying in the background of the...
- M Mathematics The volume of a sphere is given by with a radius of 4 inches. Using linear approximation, estimate the volume of the sphere if the radius is decreased by 0.04 inches....
- E English 1. when mrs. sommers leaves to go shopping, she forgets a. to take money. b. her daughter mag’s size. c. her preferred store. d. to eat lunch. 2. the resolution of a story should...
- M Mathematics I ve been having trouble with these questions over pythagorean theorem. they re questions a.-c....

Ответ:
Explanation:
Half-life of uranium = years
years
First we have to calculate the rate constant, we use the formula :
Now we have to calculate the age of the sample:
Expression for rate law for first order kinetics is given by:
where,
k = rate constant =
t = age of sample = ?
a = let initial amount of the reactant = 100
a - x = amount left after decay process =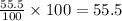
Now put all the given values in above equation, we get
Thus the age of a rock specimen that contains 55.5% of its original number of atoms is
Ответ:
The answer is B.
Explanation:
PLEASE MARK AS BRAINLIEST