ChHal3716
06.08.2021 •
Social Studies
Select a memory strategy that you think would be useful for a school-age child. On the basis of research presented in the text, what cognitive advances would enable the child to benefit from this memory strategy
Solved
Show answers
More tips
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
Answers on questions: Social Studies
- S Social Studies Ming is from china; tyrone is from canada. both participate in an experiment in which they take a test, are given feedback and told that they did very well, and then are...
- S Social Studies Summarize why 1917 is a turning point in World War 1....
- S Social Studies Which type of economy uses a central authority to make economic decisions? * A. market B. traditional C. authoritarian D. command...
- S Social Studies Dana sees no glowing embers when she looks directly at the fireplace, but when she looks a little to the side, she can see a dimly glowing spot. The best explanation for...
- S Social Studies Which of the following illustrates the concept of stereotyping? Multiple Choice Russell believes that all men love adventure. Greta considers reading a useful pastime. Laila...
- M Mathematics consider each of the roots below.if the root is a rational number ,simplify it fully .if it is an irrational number, establish the two integers it lies between.( a)√1,(b)√5,(c)√25(d)√36(e)√9÷4(f)√18÷50(g)√12+4÷4(f)√32÷4...
- B Business Please help me, who knows procedures for incorporation?. Thanks...
- M Mathematics Please answer this geometry question...
- H Health Plot the Curve and Tangent of the Following equationsy = -x + 6Y = -0.39430Cx + 2.2743...
- C Computers and Technology TRUE OR FALSE?? Binary numbers are encoded in different data formats to give them meaning, for example the 8-bit pattern 01000001 could be the number 65, or the character...

Ответ:
Rehearsal is found to be the most frequently used strategy, followed by mental imagery, elaboration, mnemonics, and organization. Previous study also found that rehearsal is the memory strategy taught most often by teachers to their students (Moely et al., 1992).
Ответ:
Explanation:
Given parameters:
Mass of the sample = 0.622g
Volume of the gas = 2.4L
Temperature = 287K
Pressure of the gas = 0.85atm
Unknown
Molar mass of the gas = ?
Solution
To find the molar mass of the gas, we first assume that the gas in question is an ideal gas i.e. it obeys Boyle's and Charles's law.
On this premise, we can find the unknown which is the molar mass of the gas.
We first use the ideal gas equation to find the number of moles of the gas since all the parameters apart from the number of moles are given:Solving for number of moles:
The ideal gas equation is given as PV = nRT
We make the unknown "n" the subject of the equation
n =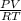
R is the gas constant whose value is 0.082atmdm³mol⁻¹K⁻¹
n =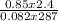
n =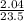 = 0.087mole
= 0.087mole
Now, we know the number of moles of the gas. We can proceed to find the molar mass from the given mass and the number of moles calculated using the equation below:molar mass =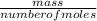
molar mass =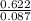 =7.15gmol⁻¹
=7.15gmol⁻¹
Part 2:
If the sample was placed under a low temperature, we want to consider what would happen to the volume:
Since we assume that the gas is an ideal gas, we can explain what will happen to the gas using Charles's law. The law states that "The volume of a fixed mass of a gas varies directly as its absolute temperature if the pressure is constant".
If we assume the pressure is constant for this gas, the volume of the gas would decrease as the temperature becomes lower. We would get lower values for both actual volume and predicted volume.