jewelia2801
01.12.2020 •
Spanish
How dose Bolsa describe Bill joe
Solved
Show answers
More tips
- F Family and Home Is It Worth Spoiling a Child?...
- S Society and Politics Will Japan become Russia s Military Enemy?...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Society and Politics Skoptsy: Who They Are and How They Perform Castration?...
- O Other Childhood Fears: What Many of Us Experienced...
- P Philosophy What is Something for you?...
- H Health and Medicine Why Do Humans Have One Heart?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
Answers on questions: Spanish
- L Law QUESTION 17 Indicate the correct statement: Court of first instance and court a quo refer to the court which has heard the case for the first time (2) The Magistrate court...
- M Mathematics Jason had $85 to spend on 9 books. after buying them, he had $13 left. how much did each book cost? write an equation and solve. meee marking as...
- P Physics Two steel guitar strings have the samelength. string a has a diameter of 0.50mm and is under 440.0 n oftension. string b has a diameter of 1.0 mm and is under a tensionof...

Ответ:
what
Explanation:
Ответ:
1M
Explanation:
Given parameters:
Volume of Ca(OH)₂ = 25mL = 0.025dm³
Concentration = 1.1M
Volume of HNO₃ = 55mL = 0.055dm³
Unknown:
Concentration of HNO₃ = ?
Solution:
To solve this problem, we must bear in mind that this an acid-base titration. In this reaction, an acid is used to neutralize a base.
The reaction equation is shown below;
Ca(OH)₂ + 2HNO₃ → Ca(NO₃)₂ + 2H₂O
Start from the given and known reactant first. Here it is Ca(OH)₂ ;
Find the number of moles of Ca(OH)₂ first.
Number of moles Ca(OH)₂ = concentration x volume
= 1.1 x 25 x 10⁻³
= 0.028moles
From the reaction equation;
1 mole of Ca(OH)₂ reacted with 2 moles of HNO₃
0.028 moles of Ca(OH)₂ will react with 2 x 0.028
= 0.055moles of HNO₃
Now that we know the number of moles, we can find the concentration of HNO₃;
Concentration of HNO₃ =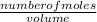
=
= 1M