1. a 5.455-g sample of impure cacl2 is dissolved and treated with excess potassium carbonate solution. the dried caco3 (calcium carbonate) precipitate weighs 4.010-g. calculate the percent by mass of cacl2 in the original mixture.
Solved
Show answers
More tips
- F Food and Cooking When is Easter in 2011?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- H Health and Medicine How to perform artificial respiration?...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
Answers on questions: Chemistry
- C Chemistry How many ounces of water should be evaporated from 90 ounces of a 10% salt solution in order to make a 15% salt solution?...
- C Chemistry The decomposition of hi(g) is represented by the equation 2hi(g)⇌h2(g) i2(g) the following experiment was devised to determine the equilibrium constant of the reaction. hi(g) is introduced...
- C Chemistry Acube with edge = 1.00” of an unknown pure substance has a mas off 10.0 oz. what is the density of this substance in g/ml? in kg/l?...
- C Chemistry Aflashbulb of volume 2.70 ml contains o2(g) at a pressure of 2.30 atm and a temperature of 30.0 °c. how many grams of o2(g) does the flashbulb contain?...
- C Chemistry Why do hot objects have high temperatures?...
- C Chemistry Can anyone draw a diagram of a liebigs condenser...
- L Law Manuela began renting her entire main home at fair rental value on May 1, 2008. When she converted the house to rental property, the adjusted basis was $205,000, which included $18,000...
- A Arts Which of these techniques did Raphael use in his portraits?OA. He highlighted features other than the face to create a sense ofmovementB. He created naturalistic backgrounds to give...
- E English Questions 1-10: Substitute contractions for the words in italics....
- H Health All of the following are part of appropriate netiquette except using all capital letters avoiding slang and informal language having a proper greeting at the beginning being sure...

Ответ:
The mass percent of calcium chloride in the sample is 81.6 %
Explanation:
To calculate the number of moles, we use the equation:
Given mass of calcium carbonate = 4.010 g
Molar mass of calcium carbonate = 100 g/mol
Putting values in equation 1, we get:
The chemical equation for the reaction of calcium chloride and potassium carbonate follows:
By Stoichiometry of the reaction:
1 mole of calcium carbonate is produced from 1 mole of calcium chloride
So, 0.0401 moles of calcium carbonate will be produced from =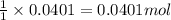 of calcium chloride
of calcium chloride
Now, calculating the mass of calcium chloride by using equation 1:
Molar mass of calcium chloride = 111 g/mol
Moles of calcium chloride = 0.0401 moles
Putting values in equation 1, we get:
To calculate the mass percentage of calcium chloride in sample, we use the equation:
Mass of sample = 5.455 g
Mass of calcium chloride = 4.451 g
Putting values in above equation, we get:
Hence, the mass percent of calcium chloride in the sample is 81.6 %
Ответ:
Explanation:
hope this helps you to understand :)