kyramks421
13.04.2020 •
Chemistry
1. If a gas at 37 degrees Celcius occupies 2.9 L at a pressure of 380 torr, what will be the new
volume at a pressure of 1.5 atm?
LAW:
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- S Style and Beauty How to Sew Harem Pants?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry 2AlBr3 + 3BaF2 → 2AlF3 + 3BaBr2 Calculate how many moles of AlBr3 would be used if 4.95 moles of BaBr2 were produced in the reaction you balanced...
- C Chemistry The graph shows the potential energy diagram for an unknown chemical change. Which chemical change could the graph represent? (1 point) Photosynthesis in plants Combustion of...
- C Chemistry Match the name of each gas law to the properties it compares. 1. Gay-Lussac s law 2. Boyle s law 3. Avogadro s law a. Volume and moles b. Pressure and volume c. Pressure and...
- C Chemistry True or false. the epicenter of the earthquakr is directly below the focus...
- C Chemistry When 1.00 mol of no( g ) forms from its elements, 90.29 kj of heat is absorbed. how kj much heat is evolved when 2.79 g of no decomposes to its elements? assistance...
- C Chemistry Classify each of the changes as a physical change or a chemical change. burning wood...
- C Chemistry How does a chemical bond form in sodium chloride? Sodium atoms an electron to form a sodium ion with a charge of Chlorine atoms __ an electron to form a chloride ion with a charge...
- C Chemistry Ome fire extinguishers contain liquid carbon dioxide Suggest how carbon dioxide...
- S Social Studies What was the result of Jordan, Egypt, Syria, Iraq, and Lebanon declaring war and invading Israel? A. Israel lost a great deal of the land that had been allocated to it. B. Israel...
- E English CAN ANYONE PLS HELP ME IN THIS PLS I WILL MARK U AS BRAINLIEST...

Ответ:
0.96 litre will be the new volume of the gas at a pressure of 1.5 atm.
Explanation:
Data given:
initial temperature of the gas = 37 degrees
initial pressure P1 = 380 torr or 0.5 atm
initial volume of gas V1= 2.9 litre
final volume V2 = ?
final pressure P2 = 1.5 atm
Assuming that temperature remains constant, the formula used is of Boyle's Law:
P1V1 = P2V2
Rearranging the equation to know the value of final volume of the gas:
V2 =
V2 =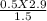
V2 = 0.96 LITRE
the volume of the gas decreases as the pressure of the gas increases.
Ответ:
Elevation
Explanation:
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation
Elevation