7letters22
09.01.2020 •
Chemistry
1. use electron dot formulas to predict the formulas of the ionic compounds formed from the
following elements:
a) beryllium and sulfur
b) potassium and fluorine
c) magnesium and oxygen
d) hydrogen and sulfur
e) aluminum and chlorine
f) iodine and sodium
g) selenium and strontium
h) gallium and oxygen
Solved
Show answers
More tips
- F Family and Home How to Teach Your Child to Read?...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- H Health and Medicine How to perform artificial respiration?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
Answers on questions: Chemistry
- C Chemistry Approximately how many moles of boron make up 1.20 × 10^24 atoms of boron? a. 7.22 × 10^47 moles b. 6.02 × 10^23 moles c. 5.00 moles d. 1.99 moles...
- C Chemistry My sna- mercedes_r624...
- C Chemistry For the following equation insert the correct coefficients that would balance the equation. If no coefficient is need please insert the NUMBER 1. 1. CH4 + O2 -- CO2...
- M Mathematics Fr pointsss.. :))And follow me for a follow backk : pleaseee♡♡...
- M Mathematics How to Subtract this and put in Standard form?...
- M Mathematics Information / is a special kind / you need / of website that can help you /find...
- M Mathematics Missy read 1/12 of a book by monday, 1/4 of the book by tuesday, and 5/12 of the book by wednesday. what is the rule for how much of the book missy reads each day? show...

Ответ:
To write the ionic formula of the compound, the cationic and anionic ions are combined with their charges.
(a) Beryllium and Sulfur:
Cation:
Anion =
The net charge = +2-2 = 0
Formula =
(b) Potassium and Fluorine
Cation =
Anion =
Net charge = 1-1 = 0
Formula = KF
(c) Magnesium and oxygen:
Cation =
Anion =
Net charge = +2 - 2 = 0
Formula = MgO
(d) Hydrogen and Sulfur :
Cation :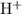
Anion :
To combine with 1 molecule of S and balance the net charge 0. 2 hydrogen ions are used.
Formula =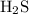
(e) Aluminium and Chloride:
Cation:
Anion :
To combine with 1 molecule of Al and balance the net charge 0. 3 Cl ions are used.
Formula =
(f) Iodine and Sodium:
Cation: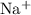
Anion:
Net charge: +1 - 1 = 0
Formula = NaI
(g) Selenium and strontium:
Cation:
Anion:
Net charge = +2 - 2 = 0
Formual = SrSe
(h) Gallium and Oxygen:
Cation:
Anion:
Net charge: 0
Formula :
For more information, refer to the link:
link
Ответ:
a) Bes
b) KF
c) MgO
d) H2S
e) AlCl3
f) NaI
g) SrSe
h) Ga2O3
Explanation:
In the picture attached the electron dot representation of different elements is shown.
a) From the picture it an be seen that Beryllium has 2 valence electrons, to form a ionic compound it will lost them and transforms into
Sulfur has 6 valence electrons, to form a ionic compound it will gain 2 electrons (to complete 8 electrons in its outer shell) and transforms into
The ionic compounds are electrically neutral, so the charge combination of its ions must be zero. If we combine 1 cation with 1
cation with 1  anion the net charge will be: +2 + (-2) = 0. Then the formula is BeS.
anion the net charge will be: +2 + (-2) = 0. Then the formula is BeS.
The other cases are solved analogously.
b)
potassium cation:
fluorine anion:
compound formula: KF (net charge: +1 + (-1) = 0)
c)
magnesium cation:
oxygen anion:
compound formula: MgO (net charge: +2 + (-2) = 0)
d) hydrogen and sulfur don't make an ionic compound, they form hydrogen sulfide which is a covalent compound
e) aluminum and chlorine form AlCl3, which is covalent.
f)
sodium cation:
iodine anion:
compound formula: NaI (net charge: +1 + (-1) = 0)
g)
strontium cation:
selenium anion: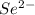
compound formula: SrSe (net charge: +2 + (-2) = 0)
h)
gallium cation: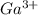
oxygen anion:
compound formula: Ga2O3 (net charge: 2*(+3) + 3*(-2) = 0)
Ответ:
The process to this is melting.
Explanation:
Due to the temperature of heating, it will melt, which will cause the solid, to a liquid.
I really hope this answers your question!