15 productos de limpieza para el hogar
Solved
Show answers
More tips
- P Philosophy Agnosticism: Opinion or Belief?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- B Biology I HAVE ASKED THREE TIMES! PLEASE HELP! CELL CYCLE! CELL DIVISION! PLEASE HELP AND GIVE REAL ANSWERS before this is due in an hour...
- M Mathematics Verna shipped three packages to chicago. if the packages weighed 12 1/2, 35 1/4, and 41 7/20 pounds, what was the total weight? a. 89 1/10 pounds b. 87 7/10 pounds c. 84 13/20...
- M Mathematics Which hypothesis or hypotheses about the relation between life satisfaction and job satisfaction has received the most empirical support...
- M Mathematics Whats the volime for this equation plzz helpp...
- M Mathematics Is it an inverse or a reciprocal? Pls help ASAP!!!...

Ответ:
We must use 5 moles of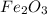
8.19 moles of oxygen are needed
Explanation:
The balanced chemical reaction is:
As is the limiting reagent as it limits the formation of product and is the excess reagent.
According to stoichiometry :
2 moles of require = 1 mole of
Thus 10 moles of will require=
will require= of
of 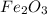
Thus we must use 5 moles of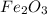
The balanced chemical reaction is:
According to stoichiometry :
2 moles of require = 3 mole of
Thus 5.46 moles of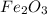 will require=
will require=
8.19 moles of oxygen are needed