cjones2941
24.11.2021 •
Chemistry
2.44 m 638cm 0.005 km 700mm smallest to largest
Solved
Show answers
More tips
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...

Ответ:
The answer to the question is
The enzyme must lower the activation energy by 35624.5 KJ/mol
Explanation:
The relation is
k =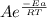
Where k = rate of chemical reaction
A = Arrhenius constant
Ea = Activation energy
T = Temperature
R = Gas constant
at 37 °C we have T = (37 +273.15) K = 310.15 K
k =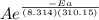
when a catalyst is introduced we have Ea(catalyst)
kc =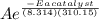
Finding the ratio of the two reaction rate and noting that Kc/K = 1.0 x 10⁶
Thus we have
Kc/k =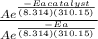 = 1.0 x 10⁶ by taking natural logarithm of both sides, we have
= 1.0 x 10⁶ by taking natural logarithm of both sides, we have
6 × ln (10) = Ln (Kc/k ) =
so that Ea - Eacatalyst = 8.314 × 310.15 × 13.816 = 35624.5 KJ/mol
Therefore the enzyme must lower the activation energy by 35624.5 KJ/molto achieve a million fold increse in the reaction rate constant