tjiaxin8082
07.03.2020 •
Chemistry
2NO + 3Fe2++ 4H2O2HNO3 + 3Fe+ 6H+ In the above reaction, the oxidation state of nitrogen changes from to . How many electrons are transferred in the reaction?
Solved
Show answers
More tips
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
Answers on questions: Chemistry
- C Chemistry According to Le Chatelier s principle, a change in pressure affects the chemical equilibrium of the reaction syst condition? The reactants and the products are in the solid...
- C Chemistry What volume of Co2 (carbon (iv) oxide)will be produced when 10g of Na2Co3(sodium trioxocarbonate (iv) reacted with excess Hcl (Hydrogen Chloride) at STP(Na=23, C=12, O=16)...
- C Chemistry Determine the new temperature of a sample of gas if the pressure changes from 0.500 atm to 0.750 atm, and the gas had an initial temperature of 315 K. The volume and number...
- C Chemistry Predict what will be observed in each experiment below predicted observation (choose one) experiment A pair of 100 mL samples of water are taken from a well bored into a large...
- C Chemistry How much energy is needed to raise the temperature of a 60g sample of aluminum from 18.5°c to 88.4°c? the specific heat capacity of aluminum is 0.897 j/g•k.[tex]q = mcδt[/tex]...
- C Chemistry How are ionic charges used in creating chemical formulas for ionic compounds?...
- E English No one provokes me with impunity. is this verbal, dramatic or situational irony. this is from the story the cast of amontillado and is basically about a amontillado the man...
- P Physics Thermal energy from the sun travels to earth by ?...
- H History Which of the following factors would best complete the chart to the right? war and violence political upheaval new freedoms in america famine and a lack of land...
- C Chemistry Awnser in the next 5 minutes correctly and i will mark u brainliest and 20 points...

Ответ:
Answer :
The oxidation state of nitrogen changes from (+2) to (+7).
The number electrons transferred in the reaction are, 6 electron.
Explanation :
Redox reaction or Oxidation-reduction reaction : It is defined as the reaction in which the oxidation and reduction reaction takes place simultaneously.
Oxidation reaction : It is defined as the reaction in which a substance looses its electrons. In this, oxidation state of an element increases. Or we can say that in oxidation, the loss of electrons takes place.
Reduction reaction : It is defined as the reaction in which a substance gains electrons. In this, oxidation state of an element decreases. Or we can say that in reduction, the gain of electrons takes place.
Rules for the balanced chemical equation in acidic solution are :
First we have to write into the two half-reactions.
Now balance the main atoms in the reaction.
Now balance the hydrogen and oxygen atoms on both the sides of the reaction.
If the oxygen atoms are not balanced on both the sides then adding water molecules at that side where the less number of oxygen are present.
If the hydrogen atoms are not balanced on both the sides then adding hydrogen ion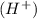 at that side where the less number of hydrogen are present.
at that side where the less number of hydrogen are present.
Now balance the charge.
The given chemical reaction is,
The oxidation-reduction half reaction will be :
Oxidation :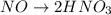
Reduction :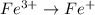
First balance the main element in the reaction.
Oxidation :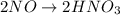
Reduction :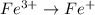
Now balance oxygen atom on both side.
Oxidation :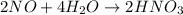
Reduction :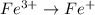
Now balance hydrogen atom on both side.
Oxidation :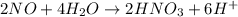
Reduction :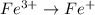
Now balance the charge.
Oxidation :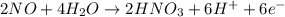
Reduction :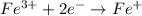
The charges are not balance on both side of the reaction. We are multiplying reduction reaction by 3 and then added both equation, we get the balanced redox reaction.
Oxidation :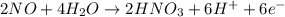
Reduction :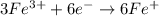
The balanced chemical equation in acidic medium will be,
The number electrons transferred in the reaction are, 6 electron.
Ответ:
I don't know the answer.
Explanation: