Anikarenee
03.06.2021 •
Chemistry
a chemist dissolves 0.564 moles of manganese (IV) oxide (MnO2) in water, and adds enough water to make 0.510 L of solution. Calculate the molarity of the solution.
Solved
Show answers
More tips
- F Food and Cooking Deflope: What is it and how does it work?...
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- F Family and Home How to Properly Use a Water Level?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
Answers on questions: Chemistry
- C Chemistry Given the reaction: 6 NaOH + 2 Al --- 2 Na3AlO3 + 3 H2 How many moles of NaOH are required to produce 3.5 moles of hydrogen?...
- C Chemistry Which phrases describe NASA s goals in the coming years? Check all that apply. 1. Improving conditions for the aviation industry 2. Conducting experiments on animals in...
- C Chemistry Photosynthesis is the process by which most living things convert the chemical energy in glucose into more accessible chemical energy in atp. a. true b. false...
- C Chemistry The particles in a gas vibrate faster than the particles in a liquid. True False...
- C Chemistry 1. Which statement best describes what Earth s outer layer is like underneath the surface in the image? a Underneath both the soil and the ocean, Earth s outer layer is...
- C Chemistry What is the name given to the way the bulbs are arranged in Diagram 1?...
- M Mathematics What is 40/3 as a mixed number...
- B Business Main cause fir collision 2 words...
- M Mathematics What is (3x7)x10 the power of 3...
- M Mathematics -4/3x=8 please step by step...

Ответ:
The molarity of the solution is 1.1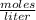
Explanation:
Molarity is a measure of the concentration of that substance that is defined as the number of moles of solute divided by the volume of the solution.
The molarity of a solution is calculated by dividing the moles of the solute by the volume of the solution:
Molarity is expressed in units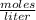
In this case
number of moles of solute= 0.564 molesvolume= 0.510 LReplacing:
Solving:
molarity= 1.1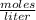
The molarity of the solution is 1.1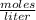
Ответ:
Explanation:
Molar mass:
Molar mass of substance is sum of atomic weight of all the atoms of elements present in it.
HCl
Atomic weight of hydrogen = 1.00 amu
Atomic weight of chlorine = 35.45 amu
Atomic weight of HCl = 1.00+35.45 = 36.45 g/mol
K₂SO4:
Atomic weight of potassium = 39.09× 2 = 78.18 amu
Atomic weight of sulfur = 32.07 amu
Atomic weight of oxygen = 15.99×4 = 63.96 amu
Atomic weight of K₂SO4 = 63.96+32.07+ 78.18 = 174.21 g/mol
Ca(OH)₂:
Atomic weight of calcium = 40.08 amu
Atomic weight of oxygen = 15.99 × 2 amu = 31.98 amu
Atomic weight of hydrogen = 1.00 × 2 = 2.00 amu
Atomic weight of Ca(OH)₂ = 74.06 g/mol
NaOH:
Atomic weight of sodium = 22.99 amu
Atomic weight of oxygen = 15.99 amu
Atomic weight of hydrogen = 1.00 amu
Atomic weight of NaOH = 39.98 g/mol
NO₃:
Atomic weight of nitrogen = 14.01 amu
Atomic weight of oxygen = 15.99×3 = 47.97 amu
Atomic weight of NO₃ = 61.98 g/mol