A sample of 2.45g aluminum oxide decomposes into 1.3g of aluminum and 1.15g of oxygen. What is the percentage composition of the compound?
Solved
Show answers
More tips
- F Food and Cooking How to Make Napoleon Cake: A Step-by-Step Guide...
- H Health and Medicine How to Increase Hemoglobin in the Blood...
- F Food and Cooking How to Make Chebureki: A Delicious Recipe for Russian Street Food...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- F Food and Cooking Is it Really Possible to Cook Tasty Colored Cauliflower?...
- H Health and Medicine Why do our Joints Crack?...
- H Health and Medicine These Food Additives Can Do Significant Harm to Your Health...
- F Food and Cooking How to Choose the Right Olive Oil: A Comprehensive Guide...

Ответ:
%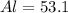 %
%
%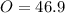 %
%
Explanation:
If we know the grams of a chemical compound in a specific reaction, it is possible to know the percentage of each atom that composes it.
For the Aluminum Oxide in this problem, we know its total weight and the grams of each component.
therefore we can determine the percentage ratio of its components through:
For Al
%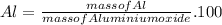 %
%
%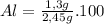 %
%
%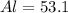 %
%
In the same way for oxygen
%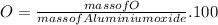 %
%
% %
%
%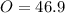 %
%
Ответ:
10.3
Explanation: