A sample of a gas occupies 2.76 L at 303K. What would the volume be if the temperature was increased to 404K? Assume constant
pressure and mass.
L Round your answer to the hundredths place.
Blank 1:
Solved
Show answers
More tips
- C Computers and Internet Are there special gaming mice?...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- C Chemistry At standard temperature and pressure, what is the volume of one mole of gas? 22.4 liters 0.0821 liters The same as the molar mass of the gas The product of protons, neutrons,...
- C Chemistry Es mas rápido el punto de ebullición añadiendo una cucharada de sal...
- C Chemistry A solution of permanganate is standardized by titration with oxalic acid, . To react completely with mol of oxalic acid required 37.02 mL of permanganate solution. The unbalanced...
- C Chemistry The frequency of the middle G note on a piano is 392.00 Hz. 392.00 Hz. What is the wavelength of this note in centimeters? The speed of sound in air is 343.06 m/s....
- C Chemistry Explain the necessary similarities and/or differences between the components of a mixture in order for extraction to be used as an effective method for separation. In other words,...
- C Chemistry PLEASE ANSWE Jazelle has five different-sized magnets. She puts each magnet far away from a paper clip and slowly moves it closer until it attracts the paper clip. She records...
- C Chemistry You have a solution of unknown concentration. You place this solution in your spectrometer and measure that it has an absorbance of 0.4 at 450 nm. The molar absorptivity of your...
- H History A- Elabora una lista de los pasos del proceso de momificación b-¿Por qué motivo era importante conservar el corazón?c-¿Por qué motivo los egipcios momificaban a sus difuntos?-...
- S Social Studies This individual was describing what historical event? 1.Bacon s Rebellion 2.French and Indian War 3.Middle Passage 4.Salem Witch Trials What is the answer...
- E English Help help it is a easy question...

Ответ:
If the temperature was increased to 404 K, its volume would be 3.68 L.
Explanation:
Charles' Law gives a relationship between the volume and the temperature of the gas at constant temperature. This law states that the volume of a given amount of gas held at constant pressure is directly proportional to the temperature.
Let
Let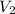 is new volume. Using above formula we get :
is new volume. Using above formula we get :
If the temperature was increased to 404 K, its volume would be 3.68 L.
Ответ:
Ans: Acetic acid = 90.3 mM and Sodium acetate = 160 mM
Given:
Acetic Acid/Sodium Acetate buffer of pH = 5.0
Let HA = acetic acid
A- = sodium acetate
Total concentration [HA] + [A-] = 250 mM (1)
pKa(acetic acid) = 4.75
Based on Henderson-Hasselbalch equation
pH = pKa + log[A-]/[HA]
[A-]/[HA] = 10^(pH-pKa) = 10^(5-4.75) = 10^0.25 = 1.77
[A-] = 1.77[HA] (2)
From (1) and (2)
[HA] + 1.77[HA] = 250 mM
[HA] = 250/2.77 = 90.25 mM
[A-] = 1.77(90.25) = 159.74 mM