A scientist produces zinc iodide (Znl2).
(a) Calculate the minimum mass of zinc that needs to be added to 0.500 g of iodine so that
the iodine fully reacts.
The equation for the reaction is:
Zn + I2 – ZnI
Relative atomic masses (Mr): Zn = 65 I = 127
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- E English In your own words, give a conclusion (small paragraph) on what you think about: why is technology important nowadays? Briefly in your own words write a paragraph explaining in detail...
- E English Is anyone down to talk...
- M Mathematics Free points. what is one of your dreams?...
- M Mathematics You deposit $1500 in a savings account earning 3.5% simple interest how much more interest would you earn if the bank compound quarterly...

Ответ:
1.24g
Explanation:
Given parameters:
Mass of titanium (IV) chloride = 4.6g
Unknown:
Mass of oxygen = ?
Solution:
To solve this problem, we need to write the balance reaction equation first;
TiCl₂ + O₂ → TiO₂ + Cl₂
Now to find the mass of the oxygen gas,
Express the given mass of the known TiCl₂ in moles:
Number of moles of TiCl₂ =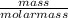
Molar mass of TiC₂ = 47.9 + 2(35.5) = 118.9g/mol
Number of moles of TiCl₂ =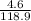 = 0.04mole
= 0.04mole
From the reaction equation;
1 mole of TiCl₂ reacted with 1 mole of oxygen gas
0.04 mole of TiCl₂ will react with 0.04 mole of oxygen gas
Mass of oxygen gas = number of moles x molar mass
molar mass of oxygen gas = 2(16) = 32g/mol
Mass of oxygen gas = 0.04 x 32 = 1.24g