19thomasar
20.12.2021 •
Chemistry
A solution is made by adding 120 g of citric acid to 210 g of water. What is the percent mass of citric acid in this solution
Solved
Show answers
More tips
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
- H Health and Medicine How to Deal with Heat Stroke?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
Answers on questions: Chemistry
- C Chemistry Which species is the conjugate base of H2SO3...
- C Chemistry Consider the molecule PF5. Indicate how many lone pairs you would find on the central atom: Indicate how many total bonds are connected to the central atom (count single...
- C Chemistry Select the correct relationship among the concentrations of species present in a 1.0 M aqueous solution of the weak acid represented by HA. A. [H2O] [HA] [A-] [H3O ] [OH-]...
- C Chemistry A 25.0 mL sample of 0.150 M hypochlorous acid is titrated with a 0.150 M NaOH solution. What is the pH at the equivalence point? The Kaof hypochlorous acid is 3.0x10^-8....
- C Chemistry A solution is prepared by dissolving 6.60 g of an nonelectrolyte in water to make 550 mL of solution. The osmotic pressure of the solution is 1.84 atm at 25 °C. The molecular...
- C Chemistry The number of organic compounds that have been reported in 2019 is more than 10 million. This is due to carbon s ability to bond to other carbon molecules, called electron...
- C Chemistry Because the double bond in an alkene is rigid, alkenes can exist as geometric isomers. To clarify geometric isomers, IUPAC uses cis- and trans- as part of a compound name....
- C Chemistry What is the pressure of the gas in problem 33,expressed in millimeters of mercury?...
- C Chemistry A 17.0 kg iron weightlifting plate has a volume of 2160 cm3 . What is the density of the iron plate in g/cm3?...
- C Chemistry 6 a. 0.9 km= mm 7 a. 11,835.76 g= Kg...

Ответ:
The concentration of neon in air is 18.14 ppm
Explanation:
We are given:
Mole fraction of neon =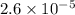
This means that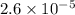 moles of neon are present
moles of neon are present
To calculate the number of moles, we use the equation:
Molar mass of neon = 20.2 g/mol
Moles of neon =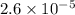 moles
moles
Putting values in above equation, we get:
ppm is the amount of solute (in milligrams) present in kilogram of a solvent. It is also known as parts-per million.
To calculate the ppm of oxygen in sea water, we use the equation:
Both the masses are in grams.
We are given:
Mass of neon =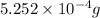
Mass of solution (air) = 28.96 g
Putting values in above equation, we get:
Hence, the concentration of neon in air is 18.14 ppm