A student mixes two solutions together. One is believed to be HI, the other is believed to be HNO3. A precipitate forms. Believing the results may be in error the student repeats the process twice in separate wells. After repeating the trial there was no observed precipitate. What is the most likely reason for the initial precipitate result?
Solved
Show answers
More tips
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- D Dating, Love, Relationships Why do we feel shame?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- P Philosophy How did the concept of module arise in computer science?...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
Answers on questions: Chemistry
- C Chemistry Use the accepted values for the molar masses of zinc and gold to determine the mass of gold needed to have the same number of atoms as 10 g of zinc....
- C Chemistry If you proceed long enough through a series of aerobic catabolic cellular reactions, ultimately what must appear as one of the products?...
- C Chemistry If a dog has a mass of 19.5 kg, what is its mass in the following units? use scientific notation in all of your answers.(a) what is the dog s mass in grams? (b) what is the dog...
- C Chemistry The empirical formula of a gaseous fluorocarbon is cf2. at a certain temperature and pressure, a 1-l volume holds 8.93 g of this fluorocarbon, whereas under the same conditions,...
- C Chemistry Which is a product of the cellular respiration reaction? O O O glucose carbon dioxide blood cells...
- C Chemistry PLEASE Which of the following is an example of an object that could have a net force greater than zero acting on it? A. a glass resting on a table B. a toy car moving east at...
- C Chemistry Should i bleach my hair ? 0.o...
- C Chemistry The most efficient way to establish the best possible economizer position is to measure...
- C Chemistry What is described in the following equation? 2H2(g) + O2(g) → 2H2O(l) A. hydrogen gas combining with oxygen gas to form water B. hydrogen gas combining with water gas to form...
- C Chemistry An electrically heated home, the temperature of the ground in contact with a concrete basement wall is 12.8 C. The temperature at the inside surface of the wall is 20.0 C. The...

Ответ:
The presence of silver nitrate impurities is the most probable cause for the initial precipitate result.
Explanation:
In test for halide ions, silver nitrate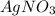 is used to test for the presence of soluble halide ions. The presence of
is used to test for the presence of soluble halide ions. The presence of 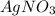 in the reaction mixture (possibly the HNO3) gave the semblance of test for chlorides, allowing for formation of precipitation in the initial experiment.
in the reaction mixture (possibly the HNO3) gave the semblance of test for chlorides, allowing for formation of precipitation in the initial experiment.
This error could have been caused by handling of reagents and used laboratory apparatus during the initial reaction.
Ответ:
0.023 moles of CaO.
Explanation:
The balanced equation tells us that we'll get 2 moles of CaO for every one mole of O2. Since we have 0.046 moles of O2, we'll get 0.023 moles of CaO. (2 sig figs)