A17.55 ml solution of potassium nitrate (kno3) was diluted to 125.0 ml, and 25.00 ml of this solution was then diluted to 1.000 × 103 ml. the concentration of the final solution is 0.00383 m. calculate the concentration of the original solution.
Solved
Show answers
More tips
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...

Ответ:
Explanation:
Given data:
V1 = 25.0 ml
M2 = 0.00383 M
V2 = 1000 ml
we knwo that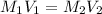
∴
= 0.1532 M
0.1532 M is concentration in 25 ml but taken from 125 ml solution.
∴ The concentration in 125.0 ml solution is = 0.1532 M
M1 = 0.1532 M
V1 = 125.0ml and V2 = 17.5.0ml
Ответ:
yes
Explanation: