andrejr0330jr
05.08.2019 •
Chemistry
A3.00-kg block of copper at 23.0°c is dropped into a large vessel of liquid nitrogen at 77.3 k. how many kilograms of nitrogen boil away by the time the copper reaches 77.3 k? (the specific heat of copper is 0.092 0 cal/g · °c, and the latent heat of vaporization of nitrogen is 48.0 cal/g.)
Solved
Show answers
More tips
- F Food and Cooking Deflope: What is it and how does it work?...
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- F Family and Home How to Properly Use a Water Level?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
Answers on questions: Chemistry
- C Chemistry Two solid samples each contain sulfur, oxygen, and sodium, only. these samples have the same color, melting point, density, and reaction with an aqueous barium chloride solution....
- S Social Studies You own a small Roman farm in the 100s B.C. Write a letter to a friend describing the changes you have witnessed in agriculture and the Roman government. Describe how those...
- S Social Studies Write the name of any 2 organization working in nepal to control corruption ?...
- M Mathematics Give a counter example to this statement. the quotient of two fractions between 0 and 1 is always a whole number....
- M Mathematics Lourdes is filling a 9-gal bucket at a rate of 0.1 gal/s. What is the domain of the function that represents the volume of water in the bucket after x seconds?...
- E English Ok who tryna talk on g00gle hangouts? warning: explicite content ppl under 14 not allowed...

Ответ:
1.2584kg of nitrogen boils.
Explanation:
Consider the energy balance for the overall process. There are not heat or work fluxes to the system, so the total energy keeps the same.
For the explanation, the 1 and 2 subscripts will mean initial and final state, and C and N2 superscripts will mean copper and nitrogen respectively; also, liq and vap will mean liquid and vapor phase respectively.
The overall energy balance for the whole system is:
The state 1 is just composed by two phases, the solid copper and the liquid nitrogen, so: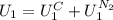
The state 2 is, by the other hand, composed by three phases, solid copper, liquid nitrogen and vapor nitrogen, so:
So, the overall energy balance is:
Reorganizing,
The left part of the equation can be written in terms of the copper Cp because for solids and liquids Cp≅Cv. The right part of the equation is written in terms of masses and specific internal energy:
Take in mind that, for the mass balance for nitrogen, ,
,
So, let's replace in the energy balance:
in the energy balance:
So, as you can see, the term disappear because
disappear because  (The specific energy in the liquid is the same because the temperature does not change).
(The specific energy in the liquid is the same because the temperature does not change).
The difference is the latent heat of vaporization because is the specific energy difference between the vapor and the liquid phases, so:
is the latent heat of vaporization because is the specific energy difference between the vapor and the liquid phases, so:
Ответ: