A50.00 ml sample of groundwater is titrated with 0.0750 m edta . if 10.30 ml of edta is required to titrate the 50.00 ml sample, what is the hardness of the groundwater in molarity and in parts per million of caco3 by mass? assume that ca2+ accounts for all of the hardness in the groundwater.
Solved
Show answers
More tips
- S Style and Beauty 5 Tips for Choosing the Best Ugg Boots...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- H Health and Medicine Effective Ways to Treat Colic in Infants...
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- C Cities and Countries What time does the Metro open in Moscow?...
- W Work and Career Where can you learn to be a flight attendant?...
- H Health and Medicine What are Electronic Cigarettes? The Ultimate Guide...
- C Computers and Internet Where are passwords stored in Opera?...
Answers on questions: Chemistry
- C Chemistry You have discovered your excel at community with people and understand whenever a person is being honest. which career might you choose ? 1) management 2)social work 3)interview 4)therapists...
- C Chemistry PLEASE ANSWERE FOR BRAINLEIST AND 100 PTS a crate on a dock weighs 300 N and covers an area of 3m 2. what pressure does the crate exert on the dock? what do you know? substitute calculate...
- C Chemistry A compound is: A. Two or more elements that are mixed together but not chemically bonded. B. A pure chemical substance consisting of one type of atom. C. None of these. D. Two or more...
- C Chemistry Many organisms in an ecosystem compete with each other for resources. What might different species of trees in a forest ecosystem compete for?...
- C Chemistry If it takes 3 hours to drive a distance of 192km on a freeway, what would be your average speed in km/h? You can use a calculator....
- M Mathematics Find a cubic function with the given zeros: 4,-6,5...
- H Health Sa mataas na markang naku 4. Alin sa mga ito ang hindi maitutring na gawi?A. PagsusugalB. Paglilinis ng ilongC. Pagpasok ng maagaD. Maalimpungatan sa gabi...
- M Mathematics A football team carried out a report to see the impact of stretching on preventing injury. Of the 50 footballers in the squad 32 stretch regularly. Of those who stretch, 4 got injured...
- M Mathematics Please solve this giving 150 points plus the crown I have been looking everywhere for this and still can t find it so please help me please odnt spam/scam me because it took me a while...
- E English What can be inferred from the information in the first paragraph? A Students are the intended audience for the text. B. Students were used as the research subjects. © Students are...

Ответ:
Concentration = 1545 ppm
Explanation:
Hardness of the water is generally due to the presence of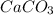 in water.
in water.
At equivalence point
Moles of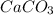 = Moles of EDTA
= Moles of EDTA
Considering
Given that:
So,
It means that 0.01545 moles are present in 1 L of the solution.
Molar mass of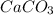 = 100 g/mol
= 100 g/mol
The formula for the calculation of moles is shown below:
Thus, moles are:
Also, 1 g = 0.001 mg
So, mass = 1545 mg
Concentration = 1545 mg/L
1 mg/L = 1 ppm
So, Concentration = 1545 ppm
Ответ:
Hey There!
To solve this question remember that 1 mole of ammonia is equal to 17G of ammonia.
Using that, we first multiply 17 by 7 = 24
Then we multiply 7 by .27 = 1.89
Finally, taking those and adding them together gives you the answer...
25.89 grams :)
Have a great day, I hope I helped.
+ Brainliest & Five stars are always appreciated