sheilashair
21.10.2019 •
Chemistry
According to the quantum-mechanical model for the hydrogen atom, which of the following electron transitions would produce light with the longer wavelength: 2p→1s or 3p→1s?
Solved
Show answers
More tips
- P Philosophy 8 привычек, чтобы достичь счастливой жизни...
- H Health and Medicine What to Eat to Lose Weight?...
- S Society and Politics Will Japan become Russia s Military Enemy?...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry Decane (c10h22) used to produce poly(ethene). what conditions are needed?...
- C Chemistry Would it be feasible to remove the copper outer layer and recover it from solution instead of the zinc core...
- C Chemistry How to solve for the net ionic equation for the reaction between aqueous solutions of nitrous acid and calcium hydroxide. How do you determine the salt?...
- C Chemistry 2H2O +O2 → 2H2O2 What mass of oxygen gas is necessary to react completely with 45 g of water?...
- C Chemistry NEED HELP ASAP EXTRA POINTS!! What type of energy would you find in a battery? A. kinetic B. sound C. thermal D. potential...
- C Chemistry What is the equation for the acid dissociation constant, Ka, of HF?...
- C Chemistry what is the boiling point of a 0.321 m aqueous solution of NaCl. Enter your rounded answer with 3 decimal places....
- C Chemistry in an experiment 3.5g of element A reacted with 4.0g of element G to form a compound Calculate the empirical formula for this compound...
- C Chemistry Determine the frequency and wavelength (in nm) of the light obsorded when the e- goes from n=1 to n=5...
- C Chemistry Determine the change in energy when an electron transitions from n= to n=5?...

Ответ:
Explanation:
The energy of transition of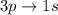 is greater as compared to
is greater as compared to 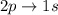 as there are more quantum levels which leads to more energy.
as there are more quantum levels which leads to more energy.
Also, considering the expression as:
Where,
h is Plank's constant having value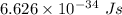
c is the speed of light having value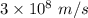
Wavelength is inversely proportional to Energy. Thus, the transition which has lower energy corresponds to higher wavelength.
Thus, answer -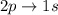
Ответ:
Inertia is based only on an object’s mass.
Explanation:
Any physical object's resistance towards any change in its velocity is known as inertia.
Therefore, more is the mass of an object, the more inertia will be there.
Thus, we can conclude that inertia is based only on an object’s mass and not on velocity.