trinwaring
21.08.2019 •
Chemistry
Aheat exchanger is to be constructed by forming copper tubing into a coil and placing the latter inside an insulated steel shell. in this exchanger, water will flow inside the tubing, and a hydrocarbon vapor at a rate of 0.126 kg/s will be condensing on the outside surface of the tubing. the inside and outside diameters of the tube are 0.0127 and 0.0152 m, respectively, inlet and exit temperatures for the water are 10 and 32°c, respectively. the heat of conden- sation of the hydrocarbon at a condensing temperature of 88°c is 335 kj/kg, and the heat- transfer coefficient for the condensing vapor is 1420 w/m².k. heat losses from the shell may be neglected. what length of copper tubing will be required to accomplish the desired heat transfer?
Solved
Show answers
More tips
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry Turn the light switch On to start the flow of current. An ammeter is a device that measures current in Amperes (A). Drag the Ammeter (A) to various parts of the circuit....
- C Chemistry The diagram illustrates photosynthesis. which best describes what is happening in the area marked y? oxygen is released into the atmosphere through stomata. carbon dioxide...
- C Chemistry A weather ballon is released into the atmosphere. You know the initial volume, temperature, and air pressure. What information will you need to predict it s volume when...
- C Chemistry What is the approximate concentration of carbon dioxide in the atmosphere? select one: 1. 380 parts per million (0.038%) 2. 7300 parts per million (0.73%) 3. 37000 parts...
- C Chemistry How many atoms of oxygen are contained in 88.2 kg of aluminum carbonate? write value in e-notation with at least three significant figures. the molar mass of aluminum...
- C Chemistry How many grams of c6h6o3 will react with 97.0grams of o2? c6h6o3+3o2 === 6co+3h2o...
- C Chemistry Carbon tetrachloride, ccl4, once used as a cleaning fluid and as a fire extinguisher, is produced by heating methane and chlorine. what mass of ch4 is needed to exactly...
- P Physics Select the best answer for the question.16. a woman named harriet miller comes before a judge in a pretrial hearing. she pleads guilty to the charge of stealing money....
- M Mathematics Abus makes a stop at 2: 30 letting on 15 people and letting on 9 the bus makes another stop in 10 minutes later to let off for more people how many more fewer people are...
- H History What was the name for the slaves of sparta?...

Ответ:
Explanation:
The given data is as follows.
Flow of mass rate of hydrocarbon, m = 0.126 kg/s
Heat of condensation, (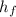 ) = 335 kJ/kg
) = 335 kJ/kg
For water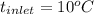 , and
, and 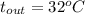
Formula for transfer of heat is as follows.
Q =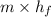
=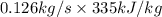
= 41.875 kJ/s
Also, it is known that Q =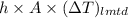
As it is given that for condensing T =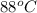 .
.
=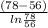
=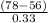
=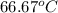
As it is given that inside and outside diameters of the tube are as follows.
Formula for A =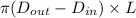 ......... (1)
......... (1)
Hence, putting value of equation (1) in the following formula.
Q =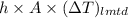
41.875 kJ/s =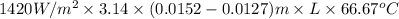
L = \frac{41.875 \times 1000 J/s}{743.17 W^{o}C}
= 56.34 h
Thus, we can conclude that length of copper tubing will be required to accomplish the desired heat transfer 56.34 h.
Ответ:
The answer to your question is below
Explanation:
Noble gases has their levels of energy completed so they can not react with another element, they can not interchange electrons .
electronic configuration
He 1 s ²
Ne 1s² 2s² 2p⁶
Ar [Ne] 3s² 3p⁶
Kr [Ar] 3d¹⁰ 4s² 4p⁶
Xe [Kr] 4d¹⁰ 5s² 5p⁶
Rn [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p⁶
In the electronic configurations we can see that Noble gases have 8 electrons in their outermost shell, so they do not need to react to be stable, they are stable.