Claysn9094
25.07.2019 •
Chemistry
Ahydrogen atom orbital has n=5 and ml=−2 .what are the possible values of l for this orbital?
Solved
Show answers
More tips
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry What are external costs? a. costs that are important in the production of goods but not always considered b. costs that form part of a product’s price c. costs that only consider...
- C Chemistry An ion of which element has a larger radius than an atom of the same element ?...
- C Chemistry Asap! which wave has the lowest amplitude?...
- C Chemistry Which of the following is NOT a correct conversion factor? 1 cm = 0.01 m 1 km = 1000 m 1 cm = 100 m 1 dm = 0.1 m ...
- C Chemistry PLEASE HELP!!! An overly enthusiastic chemistry student burns 91.00 g of propane (C3H8; 36.03 g C for each 8.07 g H) and states that 295 g of CO2 (12.01 g C for each 32.00...
- C Chemistry Help me guys please i need help...
- C Chemistry Question 65 of 99 Submit What is the correct IUPAC name for AlBrz?...
- C Chemistry Why is a comet s orbit considered a cycle?...
- C Chemistry Acertain reaction has this form: aa  bb.at a particular temperature and [a]0 = 2.00 x 10-2 molar, concentration versus time data were collected for this reaction and a plot...
- M Mathematics I need to find how many units it is?...

Ответ:
Answer : There are three possible values of 'l' which are 2, 3 and 4.
Solution : Given,
n = 5
There are 4 quantum numbers :
Principle quantum number = nvalues are 1, 2, 3, 4, .........
Azimuthal quantum number = lvalues are 0 to (n-1)
Magnetic quantum number =values are +l to -l
Spin quantum number =values are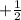 to
to 
when n = 5 then the value of l are,
l = 0, 1, 2, 3, 4
At l = 0,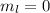
At l = 1,
At l = 2,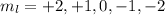
At l = 3,
At l = 4,
So, when then the possible values of l are,
then the possible values of l are,
l = 2, 3, 4
Ответ:
H-N-H bond angles : NH₂⁻ < NH₃ < NH₄⁺
Explanation:
The question tell us that the central nitrogen has four pairs of electron domains. Four pairs of electron domains predict a tetrahedral geometry but the molecular geometry will be affected by the presence of lone pair of electron and they are the ones that require more space because they have greater repulsion.
In NH₄⁺ all the 4 pairs are used in bonding with H, therefore the molecular geometry will be tetrahedral with H-N-H bond angles of 109 º.
In NH₃ we have one lone pair of electron which push the H-N-H angles less because of repulsion.
In NH₂⁻, there are two pairs of lone pairs of electrons which push the H-N-H even more.
So the order of increasing H-N-H bond angle is NH₂⁻ < NH₃ < NH₄⁺