jahjah1158
19.09.2021 •
Chemistry
Analyzing Data
Ice Cream Consumption
vs. Temperature
What conclusion can you draw
based on the data in the scatterplot?
0.55
0.45
Pints Consumed
per Person
8
0.35
.
025
20
30
T
40
50
T
60
70
80
Temperature (°F)
Done
Intro
ctivity
Solved
Show answers
More tips
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry Which of the following is true about hygroscopic substances a. they have low vapor pressures b. they remove moisture from the air c. they can be used as drying agents...
- C Chemistry The less blank, the more carbon monoxide will be produced...
- C Chemistry What is the correct lewis dot structure for h2cf2? is it linear or in box form?...
- C Chemistry ¿Cuál es la concentración molar de una solución preparada disolviendo 7 g de KOH (masa molar: 56,11g/mol) en agua y completando en matraz de aforo hasta un volumen...
- C Chemistry Rocky is a dog used to help investigate crime scenes involving fire. What is Rocky MOST likely trained to do?...
- C Chemistry Consider the following reaction: N2(g) + 3H2(g) ↔ 2NH3(g) ΔH = - 92.4 kJmol -1 a) The absolute entropy values, S, at 238 K for N2(g), H2(g) and NH3(g) are 192, 131...
- B Business Aplant asset was purchased on january 1 for $55,000 with an estimated salvage value of $5,000 at the end of its useful life. the current year s depreciation expense...
- B Business Amy s drive-thru, a fast food facility near a college campus, offers healthy, sustainably grown vegetarian and vegan fast-food at higher prices than its competitors...
- B Business Amy is single. during 2015, she determined her adjusted gross income was $12,000. during the year, amy also contributed $1,500 to a roth ira. what is the maximum...
- B Business Cincysavers prepared the following contribution margin income statement for the month. what is total contribution margin when cincysavers sells 8,200 units...

Ответ:
65.28 g of will be produced from 34.6 g of Al.
will be produced from 34.6 g of Al.
Explanation:
To calculate the moles :
The balanced chemical reaction is
According to stoichiometry :
4 moles of produce = 2 moles of
produce = 2 moles of 
Thus 1.28 moles of produce=
produce= of
of 
Mass of![[tex]Al_2O_3](/tpl/images/1174/5795/9a2b6.png) =
= 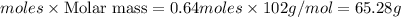
Thus 65.28 g of will be produced from 34.6 g of Al.
will be produced from 34.6 g of Al.