Which of the following is true about hygroscopic substances
a. they have low vapor pressures
b. they remove moisture from the air
c. they can be used as drying agents
d. all of the above
Solved
Show answers
More tips
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry The proton,neutron and electrons in the atom of the element represented by the symbol 231Y89 are: A. 89,231 and 89B. 142, 89 and 142C. 89, 142 and 89D. 89, 320 and 320...
- C Computers and Technology Which of the following statements is correct concerning conductivity?...
- S Social Studies Give two examples of ricks that citizens may face in a market system. Why are these risks something that would not present in a common system...
- C Chemistry If a 45-kg mass is traveling at 5m/sec and comes to a stop in 0.1 sec, how much force was applied?...

Ответ:
B is fasho the answer thank me later, It's not C because it can be used as a drying agent but not all hygroscopic substances are Drying agents and It also can be A
Ответ:
Answer : The mole fraction of methane and propane is, 0.742 and 0.26
Explanation :
First we have to calculate the moles of mixture by using ideal gas equation.
PV = nRT
where,
P = pressure of the mixture = 1.00 atm
V = Volume of the mixture = 5.04 L
T = Temperature of the mixture =![0^oC=[0+273]K=273K](/tpl/images/0505/6505/c4248.png)
R = Gas constant =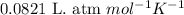
n = number of moles of mixture = ?
Putting values in above equation, we get:
Let the number of moles of methane be 'x' moles and that of propane be 'y' moles
So,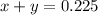 .....(1)
.....(1)
The chemical equation for the combustion of methane follows:
By Stoichiometry of the reaction:
1 mole of methane produces 1 mole of carbon dioxide
So, 'x' moles of methane will produce =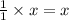 moles of carbon dioxide
moles of carbon dioxide
The chemical equation for the combustion of propane follows:
By Stoichiometry of the reaction:
1 mole of propane produces 3 mole of carbon dioxide
So, 'y' moles of propane will produce =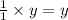 moles of carbon dioxide
moles of carbon dioxide
Now we have to calculate the mass of carbon dioxide.
Total moles of carbon dioxide = (x + 3y)
Mass of carbon dioxide = (Total moles) × (Molar mass of carbon dioxide)
Molar mass of carbon dioxide = 44 g/mol
Mass of carbon dioxide =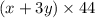
As we are given:
Mass of carbon dioxide = 15.0 g
So,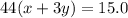 .....(2)
.....(2)
Putting value of 'x' from equation 1, in equation 2, we get:
Evaluating value of 'x' from equation 1, we get:
Mole fraction of a substance is given by:
For Methane:
Moles of methane = 0.167 moles
Total moles = 0.225
Putting values in above equation, we get:
For Propane:
Moles of propane = 0.058 moles
Total moles = 0.225
Putting values in above equation, we get:
Hence, the mole fraction of methane and propane is, 0.742 and 0.26