yasdallasj
09.07.2019 •
Chemistry
Asample of cl2 gas has a volume of 15.0 l at a pressure of 1.50 atm and a temperature of 23 °c. what volume, in liters, will the gas occupy at 3.50 atm and 286 °c? assume ideal behavior.
Solved
Show answers
More tips
- S Style and Beauty How to knit a hooded cowl?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- H Health and Medicine How to perform artificial respiration?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- F Family and Home How to Properly Use a Water Level?...

Ответ:
Given: Initial volume of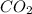 gas= 15.0 L
gas= 15.0 L
Pressure=1.50 atm
Temperature=23°C
Use ideal gas equation as follows:
Here, P is pressure, V is volume, n is number of moles, R is Universal gas constant and T is temperature in Kelvin.
First convert the given temperature in Kelvin.
0°C=273.15 K
Thus, 23 °C=296.15 K
Rearranging ideal gas equation to calculate the value of nR as follows:
Now, final pressure is 3.50 atm and temperature is 286°C, converting the temperature into Kelvin,
0°C=273.15 K
Thus, 286 °C=559.15 K
Volume can be calculated using the ideal gas equation as follows:
Putting the value of given pressure and temperature and the above calculated value of nR,
Therefore, volume of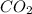 at 3.50 atm and 286°C is 12.14 L.
at 3.50 atm and 286°C is 12.14 L.
Ответ:
Volume of Cl₂ gas at 3.5 atm and 286°C = 12.14 L
Further explanationSome of the laws regarding gas, can apply to ideal gas (volume expansion does not occur when the gas is heated), among others
•Boyle's law at constant T, P = 1 / V
•Charles's law, at constant P, V = T
•Avogadro's law, at constant P and T, V = n
So that the three laws can be combined into a single gas equation, the ideal gas equation
In general, the gas equation can be written
where
P = pressure, atm
V = volume, liter
n = number of moles
R = gas constant = 0.082 l.atm / mol K
T = temperature, Kelvin
State 1
V1= 15 L
P1 = 1.5 atm
T1 = 23°C +273 = 296 K
We can find mole(n)
State 2
P2=3.5 atm
T = 286°C +273 = 559 K
n (from state 1) = 0.927

Learn moreWhich equation agrees with the ideal gas law
link
link
Which law relates to the ideal gas law
link
Keywords : ideal gas law, Cl₂
Ответ: