tylijahking
26.06.2019 •
Chemistry
Ashampoo bottle contains 553ml of air at 65c what is its volume when it cools to 22c
Solved
Show answers
More tips
- S Science and Technology Why is there no gravity on other planets?...
- F Food and Cooking Deflope: What is it and how does it work?...
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- F Family and Home How to Properly Use a Water Level?...

Ответ:
V₂ = 0.483L = 483mL
Explanation:
Given parameters:
Inital volume of air in shampoo bottle V₁ = 553mL
Original temperature of the bottle T₁ = 65°C
Final temperature T₂= 22°C
Final volume of the gas in the bottle V₂=?
We first take the given units into standard units:
K = T°C + 273
For T₁ we have the temperature to be = 65 + 273 = 338K
T₂ = 22 + 273 = 295K
For the volume we convert to Litres or dm³
1000mL = 1L
V₁ = 553 x 10⁻³L = 0.553L
Now apply one of the gas laws to solve this problem.
From Charles law, we know that the volume of fixed mass of gas is directly proportional to its absolute temperature if the pressure is constant.
Charles's law is expressed as:
The unkown in the equation is V₂ we the express it as the subject of the formula :
V₂ =
Putting the parameters in the equation gives:
V₂ =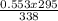
V₂ = 0.483L = 483mL
Ответ:
The correct answer is
Car A and car B are moving in opposite directions.
Car A is moving faster than car B.
Car A started ahead of car B.