At constant pressure for which of the reactions shown below should Delta H be greater than Delta E°?
1.2 SO2(g) + O2(g) - 2 SO3(g)
II. C5H12(9) + 8 O2(g) - 5 CO2(g) + 6 H20(1)
III. H2(g) + Br2(g) → 2 HBr(9),
IV. N204(9) - 2 NO2(g)
Solved
Show answers
More tips
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
Answers on questions: Chemistry
- C Chemistry Oscar s family recently put solar panels on their roof to cut down on their use of electricity from the power company how will alternative energy sources affect earth s atmosphere?...
- M Mathematics Note that GHJK has vertices G(-1, 2), H (3, - 7), J(-1, -2), and K(-5, 7). Answer the following to determine if the parallelogram is a rectangle, rhombus, square, or none of...
- M Mathematics URGENT: HELP ME WITH THE MATH PROBLEMS!! (Note This Needs To Be Done Tonight!)...
- E English Follow me on sn.ap.cha.t. a_hill2021189 btw im girl and im 15...
- M Mathematics 3 PROBLEMAS DE DECIMALES RELACIONADOS AL PROJIMO ayuda plsss...
- B Biology What effect did the changes you made it run two of the experiment when compared to run one?...
- M Mathematics An investor paid $43,000 for a lot and $520,000 to have a strip mall constructed on it. he has depreciated the property for the past 15 years on a 39-year straight-line schedule....
- S SAT Based on its context, which is the BEST meaning for the word spendthrifts as it is used in this sentence?...
- S Social Studies What were the main jobs in the Southern Colonies?...
- E English 35 PTSWhat do you write in Chapter 4 & 5 in research paper? ...

Ответ:
Explanation:
The realtion between enthalpy change and internal energy change is:
where,
= enthalpy of the reaction
= internal energy of the reaction
R = gas constant
T = temperature
1.
2.
3.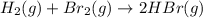
4.
Thus reaction . has higher value of
has higher value of  than
than 
Ответ:
At constant pressure the given reaction that shows ΔH would be greater than ΔE is;
IV); N₂0₄(g) → 2NO₂(g)
In chemistry, he meaning of ΔH and ΔE are;
- ΔH means the change in enthalpy, which is heat content.
- ΔE means the change in energy of a system.
Now, for a closed system the relationship between ΔH and ΔE is given by;
ΔH = ΔE + PΔV
where PΔV = ΔnRT
Thus; ΔH = ΔE + ΔnRT
where Δn = number of moles of product in the gaseous state - number of moles of reactants.
Thus, if Δn is negative, then ΔE would be greater than ΔH but if Δn is positive, then ΔH would be greater than ΔE
I) 2SO₂(g) + O₂(g) ⇒ 2SO₃(g)
number of moles of product = 2
number of moles of reactants = 2 + 1 = 3
Δn = 2 - 3
Δn = -1 and so ΔE would be greater than ΔH
II) C₅H₁₂(g) + 8O₂(g) ⇒ 5CO₂(g) + 6H₂0(l)
number of moles of product = 5 (6 is not used because water is in liquid and not gas)
number of moles of reactants = 8 + 1 = 9
Δn = 5 - 9
Δn = -4 and so ΔE would be greater than ΔH
III) H₂(g) + Br₂(g) → 2HBr(g)
number of moles of product = 2
number of moles of reactants = 1 + 1 = 2
Δn = 2 - 2
Δn = 0 and so ΔE would be equal to ΔH
IV) N₂0₄(g) → 2NO₂(g)
number of moles of product = 2
number of moles of reactants = 1
Δn = 2 - 1
Δn = +1 and so ΔH would be greater than ΔE
Read more at; link
Ответ:
this is not a question but hey add my insta plz pup.daisy.diesel I need followers